Superantigens: a brief review with special emphasis on dermatologic diseases
Published Web Location
https://doi.org/10.5070/D347g8w51mMain Content
Superantigens: a brief review with special emphasis on dermatologic diseases
Lakhan Singh Solanki MBBS, Neeraj Srivastava, MD, Sanjay Singh MD
Dermatology Online Journal 14 (2): 3
Department of Dermatology, Institute of Medical Sciences, Banaras Hindu University, Varanasi, India. sanjaye2@gmail.comA. Brief review of superantigens
1. General considerations and history
Superantigens are microbial or viral toxins that comprise a class of disease-associated, immunostimulatory molecules and act as Vβ-restricted extremely potent polyclonal T cell mitogens. They bind major histocompatibility complex (MHC) class-II molecules without any prior processing and stimulate large number of T cells (up to 20% of all T cells) on the basis of epitope specified by this receptor [1, 2, 3]. These properties are attributable to their unique ability to cross-link MHC class II and the T cell receptor (TCR), forming a trimolecular complex [1, 2]. Here we present a brief overview of superantigens with special emphasis on their role in dermatological diseases.
In 1978 Todd et al. characterized toxic shock syndrome (TSS) as a discrete clinical entity and established that it is a manifestation of staphylococcal infection [4]. Subsequently in 1981, Schlievert et al. and Bergdoll et al. [5] isolated pyrogenic exotoxin C (PEC) and staphylococcal enterotoxin F (SEF), respectively. In 1989 this fundamentally new class of antigens was recognized and termed as "superantigen" by Kappler and Marrack [6, 7].
2. Classification
The superantigens can be broadly classified into following families:
- i. Endogenous superantigens: These superantigens are encoded by various viruses integrated into the genome. Examples are superantigens produced by mouse mammary tumor virus (MMTV) and Epstein-Barr virus (EBV) associated superantigen [6, 8-11].
- ii. Exogenous superantigens: These include the exotoxins secreted by microorganisms. Examples are staphylococcal enterotoxins (A, B, C1 to C3, etc.), streptococcal pyrogenic exotoxins (A1 to A4, C, etc) and others (see Table 1) [7, 12-18].
- iii. B-cell superantigens: Those superantigens which stimulate predominantly B cells. Examples include staphylococcal protein A and protein Fv [19, 20, 21].
3. Structure
Superantigens are globular proteins synthesized as 25-30kDa precursors and secreted as 22-29kDa proteins. They are resistant to proteases and heat denaturation and are able to absorb by the epithelium as immunologically intact proteins [1]. The macromolecular structure of superantigens can be studied by X-Ray crystallography and solution nuclear magnetic resonance spectroscopy [22]. They have a common two-domain architecture (amino and carboxy terminal domains) with a long solvent accessible α-helix (part of the carboxy terminal domain) spanning the centre of the molecule [1].
The amino terminal domain consists of concave β-barrel with α-helix at one end. This domain resembles the 'oligosaccharide-/oligonucleotide-binding fold' (OB-fold) present in other protein families. In these proteins, the OB-fold is involved in DNA binding and carbohydrate recognition, respectively, but no such functions have yet been attributed to superantigens. Others features of the amino-terminal domain include the presence of several hydrophobic residues in solvent-exposed regions of the molecule, and a disulphide bridge. The residues between the two cysteines in the amino acid sequence form a highly mobile loop region [1].
The carboxy-terminal domain consists of four stranded β-sheet flanked by a long central α-helix and has some structural features of β-grasp motif present in other proteins like ubiquitin, immunoglobulin-binding domains etc. In addition, the amino-terminal tail (~15-20 residues in length) is packed against the β-grasp motif and it is considered part of the carboxy-terminal domain [1].
Most of the superantigens, except SEB, TSST-1 and SSA, possess either one or two zinc-binding sites, but the location varies in different superantigens. The zinc-binding sites seems to have a direct effect on the recognition of superantigens by MHC-class II molecules [1]. The three-dimensional structures of various superantigens are available on the internet [23].
4. Bioactivity
Superantigens, like conventional antigens/haptens, activate antigen-presenting dendritic cells by producing increased expression of HLA-DR antigen and co-stimulatory molecules (CD54, CD83 and CD86) and the production of tumor necrosis factor (TNF) -α [24, 25]. This augmentation by superantigens can be suppressed by both corticosteroids and cyclosporine, while conventional antigen/hapten-induced augmentation is resistant to suppression by cyclosporine [25].
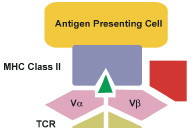
Superantigens are active at very low concentration i.e., 10mol/L [26]. Superantigen's bioactivity depends on its ability to bind both the MHC class II and TCR. Once secreted, superantigens require no processing in order to interact with the antigen-presenting cell. They interact with MHC class II molecules outside the antigen binding groove, and yet are still able to elicit a productive interaction with T cells (Fig. 1). However, differences in amino acid sequences dictate that each superantigen has an affinity for specific MHC class-II alleles [1, 6].
The MHC class-II superantigen complex is readily recognized by large families of T cells, limited only by the TCR Vβ subunit that each family expresses. Most superantigens will be recognized by at least three to five types of TCR families. After binding with T cell receptor superantigen activates large number of resting T cells, as many as 20 percent of total T cells [27]. This is followed by proliferation of T cells and their activation-induced clonal deletion. Both in vivo and in vitro superantigen-induced T cells lead to production of elevated amounts of inflammatory cytokines such as TNF-α and -β, interleukin (IL)-2, and INF-γ [1, 27]. Differences between superantigens and conventional antigens are presented in see Table 2) [6, 27-30].
5. Association of superantigens with diseases
Association of superantigens with different diseases is summarized in see Table 3 [28, 30-99].
B. Importance of superantigens in dermatology
1. Staphylococcal toxic shock syndrome
Toxic shock syndrome (TSS) is a multi-organ systemic illness due to exotoxin-producing strains of S. aureus. It is characterized by a generalized erythematous eruption and high fever. Additional elements of syndrome include hypotension, functional abnormalities in at least three organ systems, and desquamation following the scarlatiniform eruption [33].
Toxic Shock Syndrome occurs in two forms: menstrual and non-menstrual. Toxic Shock Syndrome that occurs during menstruation is associated with use of super-absorbent tampons, which cause cervicovaginal ulcerations and creates a portal for toxin absorption. Super-absorbent tampons also allow increased oxygen tension which results in increased amount of toxin production [34]. Non-menstrual TSS occurs in association with staphylococcal infections (skin, soft tissues, bone, and lung) in children, men, and non-menstruating women [33]. Recurrence usually occurs in menstrual TSS, which requires two factors, persistent colonization with toxin producing S. aureus and persistent absence of toxin neutralizing antibodies [34]. Toxin produced by menstrual-TSS associated strains of S. aureus is TSST-1. S. aureus strains of non-menstrual TSS produce TSST-1, SEB, SEC, and both TSST-1 and SEC1 in 48 percent, 26 percent, 7 percent and 19 percent of cases, respectively [35].
Superantigenic stimulation produced by these toxins lead to production of cytokines like IL-1 and TNF-α. Sudden increase in TNF-α is accompanied by changes in endothelial and vascular smooth muscle changes, which manifests as hypotension, shock, and features of sepsis [9, 27].
2. Staphylococcal scalded skin syndrome
It is a generalized exanthematous disorder with cutaneous tenderness, widespread blistering and superficial denudation. Strains of S. aureus that cause staphylococcal scalded skin syndrome (SSSS) usually belong to group II phage types 71 and 55, and sometimes to phage groups I and V [33]. The staphylococcal exfoliative toxins A and B (ETA and ETB), by their superantigenic property, act as trypsin-like serine protease [36] or act as lipase and activate other proteases [37]. They also bind directly to the desmosomal cadherin desmoglein I (DsgI), which results in disruption of desmosomes in the granular layer of epidermis, leading to interadesmosomal splitting, chacteristic blistering and denudation of skin [36, 38]. Other clinical manifestations of SSSS are not explained by exfoliative toxins (ETs), but are probably caused by δ hemolysin secreted by S. aureus, a cytolytic toxin that has detergent like effect on cell membranes [39].
3. Staphylococcal scarlatiniform eruption
It is considered a milder variant of SSSS. Staphylococci belonging to phage group II are responsible. Strains causing this eruption produce staphylococcal enterotoxin (SEs) similar to the toxins responsible for TSS [33].
4. Guttate psoriasis and psoriasis vulgaris
Psoriasis is a common, chronic, inflammatory and proliferative skin disease, in which both genetic and environmental factors play crucial roles. Newer concepts suggest that it is a Th-1-mediated disease. Evidence shows that T cells in psoriasis are triggered by conventional antigens and superantigens. It is suggested that although the process is initiated by bacterial superantigens, molecular mimicry between the bacterial antigens and keratin 17 leads to activation of autoreactive T cells and persistence of disease [40].
Acute guttate psoriasis is preceded or is concurrent with group-A streptococcal infection, particularly of throat [41, 42, 43] and is associated with rise in serum antistreptococcal titres [44]. As a result of superantigen activation, there is enhanced expression of Vβ2 cells in acute skin lesions of patients with guttate psoriasis. It was is found that there was selective expansion of Vβ2+ cells in both the CD4+ and CD8+ infiltrating T cells in the dermis and epidermis of guttate psoriasis. Superantigens involved in the activation of Vβ2+ T cells activation in guttate psoriasis were analyzed in the isolates of group A streptococcus by M typing and secretion of pyrogenic exotoxins A, B, and C [41]. Results showed that in most patients there is no consistent M protein type. Some isolates secreted SPEA, others SPEB, which could explain the expansion of Vβ8 seen in some patients. All streptococci secrete SPEC, a superantigen known to stimulate marked expansion of Vβ2+ T cells, therefore it appears to play important role in the pathogenesis of this disease [41].
To explain eruption of lesion of guttate psoriasis following streptococcal infection of throat, it has been postulated that after pharyngitis, skin-seeking T cells are induced in the lymph nodes draining the pharynx. After activation, these T cells then home to the skin via (CLA)/E-selectin interaction and may be further locally activated by a skin-specific antigen that is recognized by Vβ2+T cells. Recent evidence further supports the hypothesis that SPEC and other bacterial superantigens potentially induce the expression of skin-homing receptor cutaneous lymphocyte-associated antigen (CLA) on T lymphocytes, in an IL-12 dependent manner [41]. Patients with guttate psoriasis frequently improve with systemic antibiotic therapy [45].
A recent prospective study has confirmed anecdotal and retrospective reports that streptococcal throat infection can cause exacerbation of chronic plaque psoriasis [46].
5. Kawasaki syndrome
Kawasaki syndrome (KS) is an acute multisystem vasculitis that primarily affects infants and young children. It is widely agreed that KS is caused by an infectious agent because of the acute, self-limited nature of this disease, seasonal incidence, geographic clustering of outbreaks, and the unique susceptibility of young children. Furthermore, fever and other clinical findings in acute KS overlap with bacterial toxin mediated disease such as toxic shock syndrome (TSS) or scarlet fever. Initial few reports provided inconclusive evidence about Vβ2 expansion in acute KS [47, 48, 49].
One study done on 19 children with KS, in which TCR Vβ gene usage was assessed, found that there was marked expansion of Vβ2 and, to a lesser extent, Vβ8 in patients with acute KS. During convalescence phase of KS, their percentages reached normal level [50]. Yamashiro et al. studied 12 patients and observed the selective expansion of Vβ2-positive cells in the small intestinal mucosa in acute phase of KS. This suggests that gastrointestinal mucosa may be the primary site of entry of superantigens, produced locally by the bacteria colonizing the small intestine [51]. Potential bacterial superantigens involved in the pathogenesis were studied by Leung et al. in a blinded study [52]; they analyzed cultures of 16 patients in acute phase of KS. Superantigen producing bacteria were found in 13 of 16 patients with acute KS, but only 1 of 15 control patients (p< 0001). Of 13 toxins-positive cultures from patients with KS, 11 were TSST-1 secreting S. aureus, and 2 were streptococci, producing streptococcal pyrogenic exotoxin B (SPEB) and streptococcal pyrogenic exotoxin C (SPEC). TSST-1 and SPEC are known to posses Vβ2 stimulatory activity, whereas SPEC has both Vβ2 and Vβ8 stimulatory activity. 12 of 13 culture-positive patients had toxin-producing S. aureus isolated from the pharyngeal or rectal cultures, again suggesting gastrointestinal tract as the primary site of entry. A few new trials also suggest role of TSST-1-producing S. aureus and SPEC-producing streptococci in acute KS [52, 54]. Furthermore, there are three case reports of occurrence of psoriasis following KS suggesting the role of superantigens in KS [55, 56, 57].
6. Atopic dermatitis
Atopic dermatitis (AD) is a genetically determined, chronically relapsing, inflammatory skin disease that has a complex immunopathogenesis involving both immediate hypersensitivity and cellular responses. Superantigens fulfill Koch's postulates in AD as application of superantigen SEB to the skin induces skin erythema and induration accompanied by the infiltration of T cells that are selectively expanded in response to SEB [58, 59, 60].
Although the pathogenic role of superantigens may not be of primary importance, superantigens appear to be one of the important triggering factors that contribute to the cutaneous inflammation in AD [61]. Staphylococcus aureus is found on more than 90 percent of AD skin lesions; only 5 percent of normal individuals harbor this organism [62]. The density of S. aureus on inflamed AD lesions without clinical superinfection can reach up to 107 colony forming units per cm2. S. aureus exacerbate or maintain skin inflammation in AD through superantigens that stimulate marked activation of T cells and macrophages. The skin lesions of over half of AD patients contain S. aureus that secretes superantigens such as SEA, SEB and TSST-1 [63, 64].
Scratching disrupts the skin barrier in AD and leads to exposure of extracellular matrix molecules (fibronectin, collagen) which act as adhesins for S. aureus. IL-4 also enhances binding of S. aureus to skin by stimulating the synthesis of fibronectin [65]. In another study, bacterial binding was found to be more at the sites with Th-2 mediated inflammation [66]. Studies of peripheral blood skin-homing CLA +T cells from these patients, as well as T cells in their skin lesions, reveal that they had undergone a T cell receptor (TCR) Vβ expansion consistent with superantigenic stimulation [68, 69]. Clinical evidence for the role of S. aureus in AD includes demonstration of greater reduction in severity of lesions on treatment with combination of antistaphylococcal antibiotic and topical corticosteroid compared to topical corticosteroid alone [67].
Superantigens also induce specific IgE in AD patients and also cause mast cell degranulation in vivo, which promotes itch-scratch cycle critical to the evolution of skin rashes in AD patients. Correlation exists between presence of IgE antibodies against superantigens and severity of AD [61].
7. Cutaneous T-cell lymphoma
Cutaneous T-cell lymphoma (CTCL) is a malignancy of skin-homing T cells that are best delineated from other T cells by a unique cell surface receptor called CLA, a glycoprotein that is expressed on memory T cells that have the ability to home to skin by binding to E-selectin present on endothelial cells. Association of S. aureus with CTCL has been shown in a study in which 75 percent of patients showed positive staphylococcus culture from blood or skin, with half of these cultures positive for S. aureus carrying enterotoxin genes, such as TSST-1. The patients with positive blood cultures had Sezary syndrome or progressive plaque and tumor stage mycosis fungoides but only rarely had monoclonal expansion of a specific Vβ gene. This study suggests that S. aureus superantigen enterotoxin could provide or potentiate lymphocytic infiltration and chronic antigenic stimulation leading to clonal expansion in CTCL [72]. Other studies have also shown dominance of single Vβ family in the polyclonally expanded dermal T cell population [73].
Superantigenic stimulation of T cells is selective for cell bearing particular β chain variable (Vβ) gene segments of the T cell receptor (TCR). In humans, staphylococcal exfoliating toxin (ExT) and toxic shock syndrome toxin-1 (TSST-1) are known to stimulate Vβ-2 bearing cells; IL-1 can also act as a cofactor for stimulation of CTCL cells in combination with ExT [74]. Further studies proved the role of superantigens in leukemic phase of CTCL, by using antibodies directed against the β-chain of the T cell receptor (anti-Vβ antibodies), with increased Vβ5.1 usage in CTCL than Vβ2 usage [75]. Cutaneous colonization by S. aureus also influences the disease activity of CTCL, possibly by activation of Sezary cells by bacterial superantigenic exoproteins [76].
8. Acute juvenile pityriasis rubra pilaris
Acute juvenile pityriasis rubra pilaris is a form of pityriasis rubra pilaris (PRP) with particular clinical characteristics and course. It is usually preceded by an infectious condition and has similarities with other disorders mediated by superantigens. It has been postulated that streptococcal superantigen may have a role in the development of this disease. However, the exact nature and role of superantigen is not known [70, 71].
C. I.C. Drugs and therapies for superantigen-mediated diseases
1. Drugs currently available
i. Glatiramer acetate
Glatiramer acetate is a synthetic co-polymer of four amino acids based on the composition of myelin basic protein. It causes a significant reduction of proliferation of peripheral blood mononuclear cells (PBMC) as well as IFN-γ and TNF-α secretion. But these changes were observed in vitro, at concentration 200 microgram/ml, which is difficult to achieve physiologically [100]. Various double-blind, placebo-controlled trials have established its efficacy in relapsing-remitting multiple sclerosis and it is considered as one of the drugs of first-line choice [101].
ii. Polyclonal human intravenous immunoglobulins (IVIG)
The efficacy and safety of high dose intravenous polyspecific immunoglobulins G (IVIG) has been evaluated in multicentric, randomized, double-blind, placebo-controlled trial as an adjunctive therapy in streptococcal toxic shock syndrome as it neutralizes superantigen toxins [102]. Study in mice has shown that the IVIG preparation neutralized superantegenicity of S. pyogenes in vitro and enhances bacterial killing in a whole blood assay. When given to mice at the time of S. pyogenes infection, IVIG neutralizes circulating superantigens and reduced systemic inflammatory response. In delayed treatment settings, IVIG did not confer additional therapeutic benefit, in terms of reduction of inflammatory response, bacterial clearance or survival [103]. Different preparations of IVIG vary in their efficacy to neutralize the streptococcal superantigens, therefore there is need to optimize the type and dose of intravenous immunoglobulins used in adjunctive therapy for severe streptococcal diseases [104].
iii. Doxycycline
Pro-inflammatory cytokines mediate the toxic effect of superantigenic staphylococcal exotoxins (SE). Doxycycline inhibits SE-stimulated T cell proliferation and production of cytokines and chemokines by human peripheral blood mononuclear cells. This may explain the anti-inflammatory effects of doxycycline and its role in mitigating the pathogenic effects of SE [105].
iv. Anisodamine
Raceanisodamine hydrochloride is the active ingredient of a Chinese herbal extract that possesses the chemical structure of tropane alkaloids. It has inhibitory effect on the production of TNF-α, IL-β, IL-8 from peripheral blood monocytes stimulated with shiga toxin (Stx). Shiga toxin is the major toxin responsible for hemolytic-uremic syndrome (HUS) caused by enterohemorrhagic E. coli. Anisodamine prolongs the survival of mice injected with Stx. Study has also shown the inhibitory effect of anisodamine on activation of T cells by TSST-1 and on the release of pro-inflammatory and anti-inflammatory cytokines from TSST-1-stimulated human peripheral mononuclear cells (PBMC) [106]. It also interferes with Vβ+ T cell proliferation following injection of TSST-1, has protective effect on lethality of TSST-1 in mice, and has beneficial effects against various infections by gram-negative bacteria. This vasoactive drug has been used to treat acute disseminated intravascular coagulation in patients with bacteremic shock [106].
2. Experimental treatments
i. Pirfenadone
In vitro experiments with human peripheral blood lymphocytes revealed that pirfenidone reduced SEB-induced cytokine levels by 50-80 percent and inhibited about 95 percent of SEB-induced T cell proliferation [107]. Further study was conducted in BALB/c mice by exposing the mice with SEB, either systemically or by aerosol, and subsequently with a sublethal dose of lipopolysaccharide. In these experiments, pirfenidone given 2 to 4.5 hours after SEB resulted in 80 to 100 percent survival versus only 0 to 10 percent survival among untreated control animals. It also inhibited production of TNF-α from macrophages incubated with endotoxin and protects mice against endotoxin shock [107].
ii. Ketamine isomers
In human whole blood in vitro studies it has been shown that ketamine isomers significantly suppressed SEB-induced TNF-α production at concentrations exceeding 50 micromole [108]. Ketamine isomers at concentrations exceeding 100 micromole also significantly suppressed SEB-induced IL-6 production and at concentrations exceeding 500 micromole significantly suppressed SEB-induced IL-8 production. There is no significant differences between the effects of S(+)-ketamine and R(-)-ketamine forms [108].
iii. Triptolide
This is an oxygenated diterpene derived from a Chinese medicinal herb Tripterygium wilfordii. Triptolide inhibits SE-stimulated T cell proliferation by 98 percent and expression of IL-1β IL-6, TNF-α, INF-γ, monocyte chemotactic proteins (MIP)-1 α, MIP-1 β by human peripheral blood mononuclear cells (PBMC) [109]. It also mitigated the effect of lipopolysaccharides in dose-dependent manner.
iv. Immunoglobulin Y
Passive transfer of antibody generated in chickens (IgY) against the staphylococcal enterotoxin-B (SEB) suppressed cytokine responses and was protective in mice [110]. All rhesus monkeys treated with the IgY specific for SEB upto 4 hours after challenge survived lethal SEB aerosol exposure. These results suggest the protective role of SEB specific antibodies in non-human primates [110].
References
1. Papageorgiou AC, Acharya KR. Microbial superantigens: from structure to function. Trends Microbiol 2000; 8: 369-75.2. Acharya KR, Passalacqua EF, Jones EY, Harlos K, Staurt DI, Brehm RD. Structural basis of superantigen action inferred from crystal structure of toxic-shock syndrome toxin-1. Nature 1994; 367: 94-7.
3. Haynes BF, Fauci AS. Introduction to immune system. In: Braunwald E, Fauci AS, Kasper DL, Hauser SL, Longo DL, Jameson JL, editors. Harrison's principles of internal medicine. New York: McGraw Hill; 2005. pp. 1907-30.
4. Bonventre PF, Weckbach L, Staneck J, Schlievert PM, Thompson M. Production of staphylococcal enterotoxin F and pyrogenic exotoxin C by Staphylococcus aureus isolates from toxic shock syndrome-associated sources. Infect Immun 1983; 40: 1023-29.
5. Weckbach LS,Thompson MR, Staneck JL, Bonventre PF. Rapid screening assay for toxic shock syndrome toxin production by Staphylococcus aureus. J Clin Microbiol 1984; 20: 18-22.
6. Rodgers R, Rich RR. Antigen and antigen presentation. In: Rich RR, Fleisher TA, Shearer WT, Kotzin BL, Schroeder HW, editors. Textbook of clinical immunology: principles and practice. New York: Mosby; 2001. pp 7.1-7.17.
7. Huber BT, Hsu PN, Sutkowski N. Viral-encoded superantigens. Microbiol Rev 1996; 60: 473-82.
8. Rajagopalan G, Singh M, Sen MM, Murali NS, Nath KA, David CS. Endogenous superantigen shape response to exogenous superantigens. Clin Diagn Lab Immunol 2005; 12: 1119-22.
9. Torres BA, Kominsky S, Perrin GQ, Hobeika AC, Johnson HM. Superantigens: the good, the bad, and the ugly. Exp Biol Med 2001; 226: 164-76.
10. Choi Y, Kappler J W, Marrack P. A superantigen encoded in the reading frame of the 3' log terminal repeat of mouse mammary tumour virus. Nature 1991; 350: 203-7.
11. Tai AK, Lin M, Chang F, Chen G, Hsiao F, Sutkowski N, Huber BT. Murine Vbeta3+ and Vbeta7+T cell subset are specific targets for the HERV-K18 Env superantigen. J Immunol 2006; 177: 3178-84.
12. Leung KP, Torres BA. Prevotella intermedia stimulate the expansion of Vbeta- specific CD4 (+) Tcells. Infect Immun 2000; 68: 5420-24.
13. Acharya KR, Baker MD. Superantigen: structure-function relationships. Int J Med Microbiol 2004; 293: 529-37.
14. Thomas DY, Jarraud S, Lemercier B, Cozon G, Echasserieau K, Etienne J, Gougeon ML, Lina G, Vandenesch F. Staphylococcal enterotoxin-like toxins U2, V, two new staphylococcal superantigens arising from recombination within the enterotoxin gene toxin. Infect Immun 2006; 74: 4724-34.
15. Donadini R, Wahlberg M, Kohsaka T, Ito Y, Fields BA. Crystallization and preliminary X-ray analysis of Yersinia pseudotuberculosis-derived mitogen. Acta Crystallogr D Biol Crystallogr 2003; 59: 1330-32.
16. Zhao Y, Li Z, Drozd SJ, Guo Y, Mourad W, Li H. Crystal structure of Mycoplasma arthiritidis mitogen complexed with HLA-DR1 reveals a novel superantigen fold and a dimerized superantigen-MHC complex. Structure 2004; 12: 277-88.
17. Vasil'eva GI, Kozolovskii VN, Mishan'kin BN, Mishan'kin MB. Cholera toxin as Vibrio cholera superantigen. Zh Mikrobiol Epidemiol Immunobiol 2002; 2: 50-55.
18. Ware RE. Immune abnormalities secondary to infectious diseases. In: Rich RR, Fleisher TA, Shearer WT, Kotzin BL, Schroeder HW, editors. Textbook of clinical immunology: principles and practice. New York: Mosby; 2001. pp. 43.1-43.7.
19. Domiati-Saad R, Lipsky PE. Staphylococcal enterotoxin-A induces survival of VH3-expressing human B cells by binding to the VH region with low affinity. J Immunol 1998; 161: 1257-66.
20. Silverman GJ. B cell superantigens: possible role in immunodeficiency and autoimmunity. Semin Immunol 1998; 10: 43-55.
21. Patella V, Giuliano A, Bouvet JP, Marone G. Endogenous superallergen protein Fv induces IL-4 secretion from human Fc epsilon RI+ cells through interaction with the VH3 region of IgE. J Immunol 1998; 161: 5647-55.
22. Brunger AT, Adams PD, Clore GM, DeLano WL, Gros P, Grosse- Kunstleve RW, et al. Crystallography and NMR system: a new software suite for macromolecular structure dertermination. Acta Crystallogr D 1998; 54: 905-21.
23. RCSB Protein data bank. http://www.pdb.org/pdb/results/results.do (accessed on Mar 6, 2007).
24. Singh S, Aiba S, Manome H, Tagami H. The effects of dexamethasone, cyclosporine, and vitamin D3 on the activation of dendritic cells stimulated by haptens. Arch Dermatol Res 1999; 291: 548-54.
25. Manome H, Aiba S, Singh S, Yoshino Y, Tagami H. Dexamethasone and cyclosporin A affect the maturation of monocyte-derived dendritic cells differently. Int Arch Allergy Immunol 2000; 122: 76-84.
26. Nairn R. Immunology. In: Brooks GF, Butel JS, Morse SA, editors. Javetz, Melnick and Adelberg's medical microbiology. New York: McGraw Hill; 2004. pp. 119-45
27. Sriskandan S, Faulkner L, Hopkins P. Streptococcal pyogenes: insight into the function of the streptococcal superantigens. Int J Biochem Cell Biol 2007; 39: 12-19.
28. Scherer MT, Ignatowicz L, Winslow GM, Kappler JW, Marrack P. Superantigens: bacterial and viral proteins that manipulate the immune system. Ann Rev Cell Biol 1993; 9: 101-28.
29. White J, Herman A, Pullen AM, Kubo R, Kappler JW, Marrack P. The Vbeta-specific superantigen staphylococcal enterotoxin B: stimulation of mature T cells and clonal deletion in neonatal mice. Cell 1989; 56: 27-35.
30. Fleischer B, Schrezenmeier H. T cell stimulation by staphylococcal enterotoxins. Clonally variable response and requirement for major histocompatibility complex class-II molecules on accessory or target cells. J Exp Med 1988; 167: 1697-707.
31. Chatila T, Geha RS. Signal transduction by microbial superantigens via MHC class-II molecules. Immunol Rev 1993; 131: 43-59.
32. Harris TO, Grossman D, Kappler JW, Marrack P, Rich RR, Betley MJ. Lack of complex correlation between emetic and T-cell stimulatory activities of staphylococcal enterotoxins. Infect Immun 1993; 61: 3175-83.
33. Lee PK, Zipoli MT, Weinberg AN, Swartz MN, Johnson RA. Pyodermas: Staphylococcus aureus, Streptococcus, and other gram-positive bacteria. In: Freedberg IM, Eisen AZ, Wolff K, Austen KF, Goldsmith LA, Katz SI, editors. Fitzpatrick's dermatology in general medicine. New York: McGraw Hill; 2003. pp. 1856-78.
34. Andrews MM, Parent EM, Barry M, Parsonnet J. Recurrent nonmenstrual toxic shock syndrome: clinical manifestations, diagnosis and treatment. Clin Infect Dis 2001; 32: 1470-79.
35. Bohach GA, Fast DJ, Nelson RD, Schlievert PM. Staphylococcal and streptococcal pyrogenic toxins involved in toxic shock syndrome and related illness. Crit Rev Microbiol 1990; 17: 251-52.
36. Murono K, Fujita K, Yoshioka H. Microbiologic characterstics of exfoliative toxin producing Staphylococcus aureus. Pediatr Infect Dis 1988; 7: 313-15.
37. Ladhani S, Joannou CL, Lochrie DP, Evans RW, Poston SM. Clinical, microbial, and biochemical aspects of the exfoliative toxins causing staphylococcal scalded-skin syndrome. Clin Microbiol Rev 1999; 12: 224-42.
38. Takagi Y, Futamura S, Asada Y. Action site of exfoliative toxin on keratinocytes. J Invest Dermatol 1990; 94: 582.
39. Gemmell CG. Staphylococcal scalded skin syndrome. J Med Microbiol 1995; 43: 318-27.
40. Griffiths CEM, Camp RDR, Barker JNWN. Psoriasis. In: Burns T, Breathnach S, Cox N, Griffiths C, editors. Rook's textbook of dermatology. Oxford: Blackwell Science; 2004. pp. 35.1-35.69.
41. Leung DY, Travers JB, Giorno R, Norris DA, Skinner R, Aelion J, Kazemi LV, Kim MH, Trumble AE, Kotb M, et al. Evidence for a streptococcal superantigen-deriven process in acute guttate psoriasis. J Clin Invest 1995; 96: 1995-2106-12.
42. Zhao G, Feng X, Na A, Yongqiang J, Cai Q, Kong J, Ma H. Acute guttate psoriasis patients have positive Streptococcus hemolyticus throat cultures and elevated anti-streptococcal M6 protein titers. J Dermatol 2005; 32: 91-6.
43. Telfer NR, Chalmers RJ, Whale K, Colman G. The role of streptococcal infection in the initiation of guttate psoriasis. Arch Dermatol 1992; 128: 39-42.
44. Henderson CA, Highet AS. Acute guttate psoriasis associated with Lancefield group C and group G cutaneous streptococcal infection. Br J Dermatol 1998; 118: 559-61.
45. Rosenberg EW, Noah PW, Zanolli MD, Skinner RB Jr, Bond MJ, Crutcher N. Use of rifampicin with penicillin and erythromycin in the treatment of psoriasis. J Am Acad Dermatol 1986; 14: 761-64.
46. Gudjonsson JE, Thorarinsson AM, Sigurgeirsson B, Kristinsson KG, Valdimarsson H. Streptococcal throat infections and exacerbation of chronic plaque psoriasis: a prospective study. Br J Dermatol 2003; 149: 530-4.
47. Leung DY, Giorno RC, Kazemi LV, Flynn PA, Busse JB. Evidence for superantigen involvement in cardiovascular injury due to Kawasaki syndrome. J Immunnol 1995; 155: 5018-21.
48. Curtis N, Zheng R, Lamb JR, Levin M. Evidence for a superantigen mediated process in Kawasaki disease. Arch Dis Child 1995; 72: 308-11.
49. Pietra BA, De Inocencio J, Giannini EH, Hirsch R. TCR V beta family repertoire and T cell activation markers in Kawasaki disease. J Immunol 1994; 153: 1881-8.
50. Leung DY, Sullivan KE, Brown-Whitehorn TF, Fehringer AP, Allen S, Finkel TH, Washington RL, Makida R, Schlievert PM. Association of toxic shock syndrome toxin-secreting and exfoliative toxin-secreting Staphylococcus aureus with Kawasaki syndrome complicated by coronary artery disease. Pediatr Res 1997; 42: 268-72.
51. Yamashiro Y, Nagata S, Oguchi S, Shimizu T. Selective increase of V beta 2+ T cells in the small intestine mucosa in Kawasaki disease. Pediatr Res 1996; 39: 264-66.
52. Leung DY, Meissner HC, Fulton DR, Murray DL, Kotzin BL, Schlievert PM. Toxic shock syndrome toxin-secreting Staphylococcus aureus in Kawasaki syndrome. Lancet 1993; 342: 1385-88.
53. Leung DY, Meissner HC, Shulman ST, Mason WH, Gerber MA, Glode MP, Myones BL, Wheeler JG, Ruthazer R, Schlievert PM. Prevalence of superantigen-secreting bacteria in patients with Kawasaki disease. J Pediatr 2002; 140: 742-46.
54. Yoshioka T, Matsutani T, Toyosaki-Maeda T, Suzuki H, Uemura S, Suzuki R, Koike M, Hinuma Y. Relation of streptococcal pyrogenic exotoxin C as a causative superantigen for Kawasaki disease. Pediatr Res 2003; 53: 403-10.
55. Han MH, Jang KA, Sung KJ, Moon KC, Koh JK, Choi JH. A case of guttate psoriasis following Kawasaki disease. Br J Dermatol 2000; 142: 548-50.
56. Zvulunov A, Greenberg D, Cagnano E, Einhom M. Development of psoriatic lesions during acute and convalescent phases of Kawasaki disease. J Paediatr Child Health 2003; 39: 229-31.
57. Garty B, Mosseri R, Finkelstein Y. Guttate psoriasis following Kawasaki disease. Pediatr Dermatol 2001; 18: 507-8.
58. Strange P, Skov L, Lisby S, Nielsen PL, Baadsgaard O. Staphylococcal enterotoxin B applied on intact and atopic skin induces dermatitis. Arch Dermatol 1996; 132: 27-33.
59. Skov L, Olsen JV, Giorno R, Schlievert PM, Baadsgaard O, Leung DY. Application of staphylococcal enterotoxin B on normal and atopic skin induces up-regulation of T-cells by superantigen-mediated mechanism. J Allergy Clin Immunol 2000; 105: 820-6.
60. Savinko T, Lauerma A, Lethtimaki S, Gombert M, Mauri ML, Fyhrquist-Vanni N, Dieu-Nosjean MC, Kemeny L, Wolff H, Homey B, Alenius H. Topical superantigen exposure induces epidermal accumulation of CD8+T cells, a mixed Th1/Th2-type dermatitis and vigorous production of IgE antibodies in the murine model of atopic dermatitis. J Immunol 2005; 175: 8320-6.
61. Taskapan MO, Kumar P. Role of staphylococcal superantigens in atopic dermatitis: from colonization to inflammation. Ann Allergy Immunol 2000; 84: 3-10.
62. Leung DYM. Atopic dermatitis: new insight and opportunities for therapeutic inetervention. J Allergy Clin Immunol 2000; 105: 860-76.
63. Leung DYM, Harbeck R, Bina P, Reiser RF, Yang E, Norris DA, Hanifin JM, Sampson HA. Presence of IgE antibodies to staphylococcal exotoxin on the skin of patients with atopic dermatitis. Evidence for a new group of allergens. J Clin Invest 1993; 92: 1374-80.
64. Breuer K, Wittmann M, Bosche B, Kapp A, Werfel T. Severe atopic dermatitis is associated with sensitization to staphylococcal enterotoxin B (SEB). Allergy 2000; 55: 551-5.
65. Cho SH, Strickland I, Boguniewicz M, Leung DY. Fibronectin and fibrinogen contribute to the enhanced binding of Staphylococcus aureus to atopic skin. J Allergy Clin Immunol 2001; 108: 269-74.
66. Cho SH, Strickland I, Tomkinson A, Fehringer AP, Gelfand EW, Leung DY. Preferential binding of Staph. aureus to skin sites of Th2-mediated inflammation in a murine model. J Invest Dermatol 2001; 116: 658-63.
67. Leyden JJ, Kligman AM. The case for steroid-antibiotic combination. Br J Dermatol 1997; 96: 179-87.
68. Bunikowski R, Mielke ME, Skarabis H, Worm M, Anagnostopoulos I, Kolde G, Wahn U, Renz H. Evidence for a diseasae-promoting effect of staphylococcal aureus-derived exotoxin I in atopic dermatitis. J Allergy Immunol 2000; 105: 814-9.
69. Strickland I, Hauk PJ, Trumble AE, Picker LJ, Leung DY. Evidence for superantigen involvement in skin homing of T cells in atopic dermatitis. J Invest Dermatol 1999; 112: 249-53.
70. Betlloch I, Ramon R, Silvestre JF, Carnero L, Albares MP, Banuls J. Acute juvenile pityriasis rubra pilaris: a superantigen mediated disease? Pediatr Dermatol 2001; 18: 411-14.
71. Mohrenschlager M, Abeck D. Further clinical evidence for involvement of bacterial superantigens in juvenile pityriasis rubra pilaris (PRP): report of two cases. Pediatr Dermatol 2002; 19: 569.
72. Jachow CM, Cather JC, Hearne V, Asano AT, Musser JM, Duvic M. Association of erythrodermic cutaneous T-cell lymphoma, superantigen-positive Staphylococcus aureus, and oligoclonal T-cell receptor VB gene expression. Blood 1997; 87: 32-40.
73. Linnemann T, Gellrich S, Lukowsky A, Mielke A, Audring H, Sterry W. Polyclonal expansion of T-cells with the TCR V beta type of the tumour cell in lesions of cutaneous T-cell lymphoma: evidence for possible superantigen involvement. Br J Dermatol 2004; 150: 1013-7.
74. Tokura Y, Heald PW, Yan SL, Edelson RL. Stimulation of cutaneous T-cell lymphoma cells with superantigenic staphylococcal toxins. J Invest Dermatol 1992; 98: 33-7.
75. Vonderheid EC, Boselli CM, Conroy M, Casaus I, Espinoza LC, Venkataramani P, Bigler RD, Hou JS. Evidence for restrictive V beta usage in the leukemic phase of cutaneous T cell lymphoma. J Invest Dermatol 2005; 124: 651-61.
76. Tokura Y, Yagi H, Ohshima A, Kurokawa S, Wakita H, Yokote R, Shirahama S, Furukawa F, Takigawa M. Cutaneous colonization with staphylococci influences the disease activity of Sezary syndrome: a potential role for bacterial superantigens. Br J Dermatol 1995; 133: 6-12.
77. Conley DB, Tripathi A, Seiberling KA, Schleimer RP, Suh LA, Harris K, Paniagua MC, Grammer LC, Kern RC. Superantigen and chronic rhinosinusitis: skewing of T-cell receptor V β-distributions in polyp-derived CD4+ and CD8+ T cells. Am J Rhinol 2006; 20: 534-9.
78. Liu T, Wang BQ, Zheng PY, He SH, Yang PC. Rhinosinusitis derived staphylococcal enterotoxin B plays a possible role in pathogenesis of food allergy. BMC Gastoenterology 2006; 6: 24.
79. Hashimoto T, Takishita M, Kosaka M, Sano T, Matsumoto T. Superantigens and autoantigens may be involved in the pathogenesis of gastric mucosa-associated lymphoid tissue lymphoma. Int J Hematol 2001; 74: 197-204.
80. Garson J, Creange A, Dolei A, Ferrante P, Jouvin-Marche E, Marche PN, Reiger F, Ruprecht K, Saresella M, Sotgiu S, Tedder R, Perron H. MSRV, synectin and the role of endogenous retroviral proteins in demyelination. Mult Scler 2005; 11: 246-50.
81. Ochi H, Osoegava M, Murai H, Minohara M, Taniwaki T, Kira J. Presence of IgE antibodies to bacterial superantigens and increased IL-13 producing-T cells in myelitic patients with atopic diathesis. Int Arch Allergy Immunol 2004; 134: 41-48.
82. Schiffenbauer J, Johnson HM, Butfiloski E, Wegrzyn L, Soos JM. Staphylococcal enterotoxins reactivate experimental allergic encephalomyelitis. Proc Natl Acad Sci USA 1993; 90: 8543-6.
83. Kraft M, Filsinger S, Kramer KL, Kabelitz D, Hansch GM, Schoels M. Synovial fibroblasts as target cells for staphylococcal enterotoxin-induced T cell cytotoxicity. Immunol 1998; 93: 20-5.
84. Shio MT, Ribeiro-Dias F, Timenetsky J, Jancar S. PAF is involved in the Mycoplasma arthritidis superantigen-triggering pathway for iNOS and Cox-2 expression in murine peritoneal cells. Exp Cell Res 2004; 298: 296-304.
85. Sicat J, Sutkowski N, Huber BT. Expression of human endogenous retrovirus HERV-K18 superantigen is elevated in juvenile rheumatoid arthritis. J Rheumatol 2006; 22: 1821-31.
86. Evereklioglu C. Current concepts in the etiology and treatment of Bechet disease. Surv Ophthalmol 2005; 50: 297-350.
87. Hirohata S, Hashimoto T. Abnormal T cell responses to bacterial superantigens in Bechets's disease (BD). Clin Exp Immunol 1998; 112: 17-24.
88. Popa ER, Stegeman CA, Kallenberg CGM, Tervaert JWC. Staphylococcus aureus and Wegener's granulomatosis. Arthritis Res 2002; 4: 77-9.
89. Stegeman CA, Cohen Tervaert JW, Slutier WJ, Manson WL, deJong PE, Kallenberg CGM. Association of chronic nasal carriage of S. aureus and higher relapse rate in Wegener's granulomatosis. Ann Int Med 1994; 120: 12-7.
90. Hamidou M, Belizna C. Superantigens and vasculitis. Ann Med Interne (Paris) 2003; 154: 96-100.
91. Kraine MR, Tisch RM. The role of environmental factors in insulin-dependent diabetes mellitus: A unsolved issue. Environ Health Perspect 1999; 107: 777-81.
92. Azar GA, Thibodeau J. Human endogenous retrovirus IDDMK(1, 2)22 and mouse mammary tumor virus superantigens differ in their ability to stimulate murine T cell hybridomas. Immunol Lett 2002; 81: 87-91.
93. Pied S, Voegtle D, Marussig M, Renia L, Miltgen F, Mazier D, Cazenave PA. Evidence for superantigenic activity during murine malaria infection. Int Immunol 1997; 9: 17-25.
94. Goodier M, Fey P, Eichmann K, Langhorne J. Human peripheral blood gamma delta T cells respond to antigens of Plasmodium falciparum. Int Immunol 1992; 4: 33-41.
95. Ohmen JD, Barnes PF, Grisso CL, Bloom BR, Modlin RL. Evidence for a superantigen in human tuberculosis. Immunity 1994; 1: 35-43.
96. Ohmen JD, Modlin RL. Evidence for a superantigen in the pathogenesis of tuberculosis. Springer Semin Immunopathol 1996; 17: 375-84.
97. Sutkowski N, Palkama T, Ciurli C, Sekaly RP. An Epstein-Barr virus-associated superantigen. J Exp Med 1996; 184: 971-80.
98. Hsiao FC, Lin M, Tai A, Chen G, Huber BT. Cutting edge: Epstein-Barr virus transactivates the HERV-K18 superantigen by docking to the human complement receptor 2 (CD21) on primary B cells. J Immunol 2006; 177: 2056-60.
99. Torres BA, Tanabe T, Yamamoto JK, Johnson HM. HIV encodes for its own CD4 T cell superantigen mitogen. Biochem Biophy Res Commun 1996; 225: 672-78.
100. Schmidt S, Balzer S, Nullen C, Schmidt M, Pohl C, VonRucker A, Klockgether T. Immunomodulatory effects of glatiramer acetate on superantigen-and mitogen-induced T-cell stimulation in vitro. Mult Scler 2002; 8: 307-9.
101. Perumal J, Filippi M, Ford C, Johnson K, Lisak R, Metz L, Tselis A, Tullman M, Khan O. Glatiramer acetate therapy for multiple sclerosis: a review. Expert Opin Drug Metab Toxicol 2006; 2: 1019-29.
102. Derenberg J, Ihendyane N, Sjolin J, Aufwerber E, Haidl S, Follin P, Anderson J, Norrby-Teglund A. Intravenous immuloglobulin G therapy in streptococcal toxic shock syndrome: a European randomized, double-blind, placebo-controlled trial. Clin Infect Dis 2003; 37: 333-40.
103. Sriskandan S, Ferguson M, Elliot V, Faulkner L, Cohen J. Human intravenous immunoglobulin for experimental streptococcal toxic shock: bacterial clearance and modulation of inflammation. J Antimicrob Chemother 2006; 58: 117-24.
104. Schrage B, Duan G, Yang LP, Fraser JD, Proft T. Different preparations of intravenous immunoglobulin vary in their efficacy to neutralize streptococcal superantigens: implications for treatment of streptococcal toxic shock syndrome. Clin Infect Dis 2006; 43: 743-46.
105. Krakauer T, Buckley M. Doxycycline is anti-inflammatory and inhibits staphylococcal exotoxin-induced cytokines and chemokines. Antimicrob Agents Chemother 2003; 47; 3630-33.
106. Nakagawa S, Kushiya K, Taneike I, Imanishi K, Uchiyama T, Yamamoto T. Specific inhibitory action of anisodamine against a staphylococcal superantigenic toxin, toxic shock syndrome toxin-1 (TSST-1), leading to down-regulation of cytokine production and blocking of TSST-1 toxicity in mice. Clin Diagn Lab Immunol 2005; 12: 399-408.
107. Hale ML, Margolin SB, Krakauer T, Roy CJ, Stiles BG. Pirfenidone blocks the in vitro and in vivo effects of staphylococcal enterotoxin B. Infect Immun 2002; 70: 2989-94.
108. Kawasaki C, Kawasaki T, Ogata M, Nandate K, Shigematsu A. Ketamine isomers suppress superantigen-induced proinflammatory cytokine production in human whole blood. Can J Anaesth 2001; 48: 819-23.
109. Krakauer T, Chen X, Howard OM, Young HA. Triptolide attenuates endotoxin and staphylococcal exotoxin-induced T cell proliferation and production of cytokines and chemokines. Immunopharmacol Immunotoxicol 2005; 27: 53-66.
110. LeClaire RD, Hunt RE, Bavari S. Protection against bacterial superantigen staphylococcal enterotoxin B by passive vaccination. Infect Immun 2002; 70: 2278-81.
© 2008 Dermatology Online Journal