A principal case of multiple lymphoid collision tumors involving both B-cell chronic lymphocytic leukemia and metastatic malignant melanoma
Published Web Location
https://doi.org/10.5070/D33vn9m70wMain Content
A principal case of multiple lymphoid collision tumors involving both B-cell chronic lymphocytic leukemia and metastatic malignant
melanoma
Aaron S Cantor1, Stergios Moschos MD2, Drazen M Jukic MD PhD3
Dermatology Online Journal 16 (7): 6
1. Department of Dermatology, University of Pittsburgh School of Medicine2. Department of Medicine, University of Pittsburgh Cancer Institute
3. Departments of Dermatology and Pathology, University of Pittsburgh School of Medicine. jukicdm@ovi.com
Pittsburgh, Pennsylvania
Abstract
B-cell chronic lymphocytic leukemia/small lymphocytic B-cell lymphoma (CLL/B-SLL) is a neoplasm of B-cell lymphocytes that occurs frequently in the older population as an asymptomatic elevation of the white blood cell count (WBC) and has a good overall prognosis. Malignant melanoma of the skin is a neoplasm derived from cutaneous melanocytes that frequently arises among the elderly and, depending on certain histopathologic features, may metastasize loco-regionally or distally. However, only one report describes synchronous presentation of these two malignancies within the same lymph node. In this report, we present the unique case of an 87-year-old male with a presumed history of indolent CLL/B-SLL, in which metastatic malignant melanoma and CLL/B-SLL both involved 112 of 145 dissected regional lymph nodes. Possible explanations regarding the mechanisms that can lead to this rare presentation of both CLL/B-SLL and melanoma in the same lymph nodes are discussed.
Introduction
Population-based studies and recent reports suggest an increased risk of B-cell chronic lymphocytic leukemia (CLL/B-SLL) following a diagnosis of cutaneous malignant melanoma (MM) [1, 2, 3, 4] that has metastasized. Even though approximately 96,000 older Americans currently live with CLL/B-SLL, the simultaneous diagnosis of MM and CLL/B-SLL malignancies is rare [5, 6]. To our knowledge, only one case of a synchronously diagnosed lymph nodal collision tumor involving metastatic MM and CLL/B-SLL has been described [7]. Our case is the first in which MM has metastasized into multiple lymph nodes within different nodal basins, most of which were involved by CLL/B-SLL (a total of 112 lymph nodes were involved by both MM and CLL/B-SLL out of 145 removed).
Case report
An 87-year-old white male with no family history of neoplasia but a 10-year personal history of peripheral leukocytosis (peripheral blood count of 96.8 to 49.2 x109/L) and lymphocytic predominance (100.0% to 94.0%), presumed to be CLL/B-SLL, developed asymptomatic cervical lymphadenopathy over a seven-month period. Medical attention was only sought for his increasing hearing problems. Physical examination by the otolaryngologist revealed 2 enlarged lymph nodes, a 5 cm painless nodule of the right neck and a 1.3 cm nodule of the left posterior neck. At the same time, an 8 mm slightly-raised non-ulcerated pigmented lesion with associated satellitosis on the vertex of his scalp was also noted.
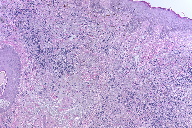
The patient underwent a punch biopsy followed by excision of the scalp skin lesion. This revealed an invasive MM with a Breslow thickness of 8.4 mm and a Clark level V (Figure 1) with an overlying ulcer and identifiable angiolymphatic invasion. Although the excision margins were not involved, multiple microsatellites were noted. Given the location of the lymphadenopathy in relation to the primary melanoma, a positron emission tomography-computed tomography scan (PET-CT scan) was performed to determine if the MM was loco-regional or distally metastatic. The CT revealed axillary, mediastinal, periaortic, and even cervical lymphadenopathy; however, only cervical lymphadenopathy was metabolically active (standard uptake value was 15.2 for right and 5.8 for left cervical lymph node regions), which suggested possible regional metastatic MM.
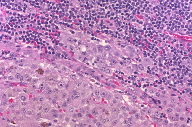
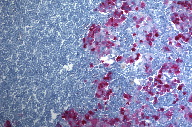
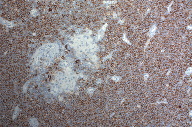
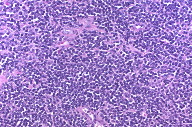
Next, the patient underwent a right and then a left modified radical neck dissection with split thickness skin grafting. The right modified radical neck dissection showed MM involving 84 percent (83 of 99) of the lymph nodes (Figures 2 and 3); the deposits of MM were positive for several markers, including S100, HMB-45, tyrosinase (Figure 4), Melan-A, and MITF. Importantly, lymphoid cells with an immunoprofile positive for CD20 (Figure 5), CD23, and CD79a and negative for CD10 and cyclin D1 involved almost 100 percent (95 of 99) of the dissected lymph nodes, which confirmed the concurrent diagnosis of CLL/B-SLL. The left modified radical neck dissection also showed MM involving 63 percent (29 of 46) of the lymph nodes (Figure 2), with the CLL/B-SLL involving 100 percent of the lymph nodes (Figure 6). Given the patient’s radiographic lymphadenopathy and lack of PET activity, it was concluded that the rest of his lymphadenopathy was predominantly CLL/B-SLL; his disease was therefore staged as stage I (Rai)/stage B (Binet) CLL/B-SLL [8].
Shortly after his second surgery, the patient developed new in-transit melanoma on his scalp. A repeat CT scan failed to reveal any new metastatic disease. The patient was later admitted to the hospital for acute renal failure that was precipitated by dehydration and hyperuricemia. Unfortunately, the patient rapidly went into multi-organ failure after readmission to the hospital and died shortly thereafter. The family chose not to have an autopsy performed.
Discussion
Patients with malignant neoplasms, such as MM, have an increased risk for developing metachronous secondary malignancies, especially CLL/B-SLL [1, 2, 3, 4]. In 2001, Cahill et al. reported that patients with MM had a standardized incidence ratio (SIR), namely the ratio of the observed over the expected cases, of 1.9 for later development of CLL/B-SLL [3]. In a study of 27,258 people identified from the Scottish national cancer registry, those diagnosed with MM had a 130 percent greater risk of subsequently developing CLL/B-SLL (SIR 2.3), in which males were at greater risk than females (SIR 2.9) and people over 50 years of age were at greater risk than younger cohorts (SIR 2.2) [1]. These high-risk features relating to coexisting malignancies, gender, and age all characterize our patient. Even though our case represents an example of the increasing association between MM and CLL/B-SLL that has been supported by several authors, it is also unique in that no other case has been reported that describes both MM and CLL/B-SLL involving many of the same lymph nodes [4, 9].
Several possible factors may explain increased rates of concurrent MM with CLL/B-SLL, as well as other non-Hodgkin lymphomas. First, the improvement in disease recording as reflected by increased clinical surveillance and greater amounts of compiled patient information has allowed for a more accurate analysis of malignant associations, but it does not provide causal links [3]. Second, cells from both MM and CLL/B-SLL share several genetic abnormalities on chromosome 9p21, such as changes in the INK4 locus that contains p16, an essential tumor suppressor protein that inhibits the cyclin dependent kinase CDNK4, which is required for cell cycle progression during G1 phase [2, 3]. This possibility was not tested in our case. Third, patients with immunodeficiencies, acquired or iatrogenic, commonly develop MM and CLL/B-SLL [3, 9] but in a manner that is possibly secondary to the defective host lymphocyte-macrophage response [2, 4]. Fourth, environmental factors have also been proposed to increase the incidence of MM and several other lymphomas. Environmental factors include exposure to chemicals, such as pesticides, herbicides, and hair dyes [3], and also exposure to the 280-315 nm wavelengths of sunlight (ultraviolet B). Ultraviolet light is thought to decrease skin hapten reactivity, inhibit proper antigen presentations, and reduce T-cell activity, all of which result in a greater risk of developing lymphoid and cutaneous malignancies [2, 3, 9].
In regard to our case, an amalgamation of these factors that stem from both MM and CLL/B-SLL etiologies could have caused our patient’s concomitant malignancies. It should also be noted that, in some elderly patients, CLL may present as a subclinical manifestation. During the period in which sentinel lymph node biopsies are performed for metastatic melanoma, the workup necessary to detect CLL is usually not initiated and, therefore, this disease can be overlooked. Thus, the incidence of presentation of both MM and CLL may be higher than the reported value in the elderly population.
Although several authors report cases of synchronously or metachronously diagnosed MM and CLL/B-SLL, El Demellawy possibly presents the first known case describing a patient with a collision tumor, in which metastatic MM and CLL/B-SLL were both present within a single lymph node [7]. Despite the similarities shared by our case and the case described by El Demellawy et al., there are two critical differences that make our case unique. First, our patient presented with multiple lymph nodes that were simultaneously involved by metastatic MM and CLL/B-SLL, whereas only a single lymph node was involved in the case of El Demellawy et al. Also, our patient had a past medical history of asymptomatic leukocytosis, whereas the patient of El Demellawy et al. had a past medical history of two MM lesions along with fevers and dramatic weight loss. Regardless of their differences, both cases serve as primary examples that mark the newly recognized risk of developing secondary malignancies following a diagnosis of either MM or CLL/B-SLL.
References
1. McKenna DB, Stockton D, Brewster DH, Doherty VR. Evidence for an association between cutaneous malignant melanoma and lymphoid malignancy: a population-based retrospective cohort study in Scotland. Br J Cancer. 2003 Jan;88(1):74-8. [PubMed]2. McKenna DB, Doherty VR, McLaren KM, Hunter JA. Malignant melanoma and lymphoproliferative malignancy: is there a shared aetiology? Br J Dermatol. 2000 Jul;143(1):171-3. [PubMed]
3. Cahill RA, McGreal G, Neary P, Redmond HP. Synchronous high-risk melanoma and lymphoid neoplasia. Melanoma Res. 2001 Oct;11(5):517-22. [PubMed]
4. Schmid-Wendtner MH, Lebeau A, Sander CA, Volkenandt M, Emmerich B, Wendtner CM. Lymphadenopathy detected by ultrasound examination as first diagnostic hint of chronic lymphocytic leukaemia in a patient with melanoma. J Eur Acad Dermatol Venereol. 2002 Sep;16(5):491-3. [PubMed]
5. Chronic Lymphocytic Leukemia, Leukemia & Lymphoma Society. 2010. [Accessed on 2010 April 5]
6. Surveillance Epidemiology and End Results, National Cancer Institute. 2010. [Accessed on 2010 April 15]
7. El Demellawy D, Ross C, Sur M, Alowami S. Synchronously diagnosed lymph nodal collision tumor of malignant melanoma and chonic lymphocytic leukemia/small lymphocytic lymphoma: case report. Diagn Pathol. 2007 Aug;2:34. [PubMed]
8. Hallek M, Cheson BD, Catovsky D, et al. Guidelines for the diagnosis and treatment of chronic lymphocytic leukemia: a report from the International Workshop on Chronic Lymphocytic Leukemia updating the National Cancer Institute-Working Group 1996 guidelines. Blood. 2008 Jun;111(12): 5446-56. [PubMed]
9. Adami J, Frisch M, Yuen J, Glimelius B, Melbye M. Evidence of an association between non-Hodgkin's lymphoma and skin cancer. BMJ. 1995 Jun 10;310(6993):1491-5. [PubMed]
© 2010 Dermatology Online Journal