Cutaneous sarcoidosis and malignancy: An association between sarcoidosis with skin manifestations and systemic neoplasia
Published Web Location
https://doi.org/10.5070/D33sh1s1qdMain Content
Cutaneous sarcoidosis and malignancy: An association between sarcoidosis with skin manifestations and systemic neoplasia
Doru T Alexandrescu MD1,2, C Lisa Kauffman MD1, Thomas E Ichim PhD3, Neil H Riordan PhD3, Filamer Kabigting MD2, Constantin A Dasanu MD PhD4
Dermatology Online Journal 17 (1): 2
1. Georgetown Dermatology, Washington, DC. mddoru@hotmail.com2. Medical School, University of California at San Diego, San Diego, California
3. Medistem Inc, La Jolla, California
4. Department of Hematology-Oncology, St. Francis Hospital and Medical Center, Hartford, Connecticut
Abstract
BACKGROUND: Whereas the association between multisystem and pulmonary sarcoidosis and malignancy has been documented, a relationship between cutaneous sarcoidosis and neoplasia has not yet been reported. Because cutaneous manifestations are seen in 20-25 percent of cases of sarcoidosis, this association deserves further investigation. METHODS: We reviewed the relevant literature, in addition to our case series, for a total of 110 cases of cutaneous and non-cutaneous sarcoidosis associated with malignancy with the aim of analyzing possible associations between cutaneous sarcoidosis and malignancy and to enhance the dermatologist’s understanding of their critical role in the management of this disease. A search for consecutive cases, which were encountered during the past 20 years, identified 10 cases of confirmed cutaneous sarcoidosis. A review of the relevant literature was also conducted to identify cases of malignancy associated with cutaneous and non-cutaneous sarcoidosis. RESULTS: Cutaneous localization of sarcoidosis was identified in 58 of 100 patients with sarcoidosis and cancer found in the literature (58%) and in 4 of 10 patients in our series (40%). In our series, all cases manifested solid tumors, including breast (n=4 tumors), prostate cancer, colon cancer, kidney cancer, and squamous cell carcinoma of the skin (n=1 of each type). Among the 6 patients in our series with cancers and non-cutaneous sarcoidosis, the types of neoplasias encountered were renal cancer (n=1), mycosis fungoides (n=1), diffuse large B-cell lymphoma (n=1), colon cancer (n=1), and ADK of parotid (n=2). Neoplasias developed after an average of 7.14 years in the literature cases and eight years in our series, following the diagnosis of sarcoidosis. Among the 100 cases of cutaneous (n=58) and non-cutaneous (n=42) sarcoidosis associates with malignancy, which were extracted from the literature, hematologic malignancies accounted for 73 percent of cases and sarcoidosis preceded the detection of neoplasia in a majority (76%) of cases. Among 110 total cases analyzed in this paper, cutaneous sarcoidosis was confirmed in 56.4 percent of overall cases, a figure exceeding expected rates of cutaneous involvement (20-25%) in the general sarcoidosis population. CONCLUSIONS: Sarcoidosis with cutaneous manifestations appears to be associated with malignancy, possibly at a higher rate than other systemic forms of sarcoidosis. The predominant occurrence of sarcoidosis before the development of neoplasia may indicate that an immune dysregulation, such as impairment of cellular immunity mediated by sarcoidosis or the effects of treatment may contribute to an increased risk of malignancy in predisposed individuals. Physician recognition of this link between sarcoidosis and malignancy is critical. Dermatologists, in particular, play an important role, given that many of these associated cases manifest initially, or even solely, with cutaneous findings.
Introduction
Sarcoidosis is a chronic idiopathic systemic disease characterized by infiltration with non-caseating granulomas. Nearly any organ system can be involved in this disease, including the liver, spleen, parotid glands, bones, joints, and central nervous system. However, the lungs, lymphatic system, eyes, and skin are most frequently affected [1, 2]. Pulmonary manifestations occur in a majority (90%) of cases [3]. Skin involvement is found in 20-25 percent of cases and commonly emerges at the onset of disease [1, 2, 4, 5]. However, cutaneous sarcoidosis may also occur in isolation in 9-30 percent of cases [4, 5, 6, 7].
Skin findings can present at any stage of systemic sarcoidosis, especially at the onset of disease; oftentimes they represent the presenting symptom [4, 5, 8, 9]. Dermatologists may be the first to evaluate the patient [4]. Therefore, any individual who presents with granulomatous skin lesions warrants a screening for systemic sarcoidosis, which should include a complete history and physical examination, chest x-rays, pulmonary function testing, electrocardiography, ophthalmologic assessment, and laboratory testing for baseline calcium, renal function, and hepatic function [8]. Subsequent screenings are critical, since isolated cutaneous sarcoidosis can develop into systemic disease in approximately one-third of patients within three years [2]. Sarcoidal skin involvement is diverse and lesions may present with many morphological varieties including: papules, plaques, nodules, lupus pernio, scar sarcoidosis, and more rare morphologies with nail involvement, ulceration, alopecia, hypopigmentation, lichenoid features, psoriasiform changes, and ichthyosis [10]. Skin lesions of cutaneous sarcoidosis are categorized into two general groups: specific and nonspecific. Nonspecific lesions lack histological evidence of non-caseating granulomas.
Erythema nodosum (EN) is most commonly seen [9], and classically presents as tender, subcutaneous nodules occurring on the anterior tibia, which are often, associated with systemic symptoms like fever, malaise, and polyarthralgias. EN is considered a hallmark of acute and benign disease [2, 11] that may represent a hypersensitivity reaction to unidentified stimuli, including medications and infection. Specific cutaneous lesions are found in an estimated 9-37 percent of patients with sarcoidosis [2, 4, 12, 13, 14]. Although morphologically diverse, these lesions share histological evidence of non-caseating granulomas. Specific lesions include: maculopapular eruptions, infiltrated plaques, lupus pernio, and scar sarcoidosis [2, 9, 12, 15, 16]. Papules are most commonly seen [2, 16, 17].
Sarcoidosis has been observed in association with various entities, ranging from autoimmune disorders, Crohn disease, celiac disease, and amyloidosis, but its association with internal malignancies has been a particular focus of intense investigation. Cases of sarcoidosis diagnosed before, concomitant with, and after the detection of cancer are well documented in the literature, but the precise nature and strength of this association remains controversial. A link between sarcoidosis and malignancy was initially proposed over three decades ago in a seminal study by Brincker et al. Patient matching, between the Danish sarcoid registry (between 1962-1971) and the Danish Cancer Registry, showed a statistically significant, higher incidence of malignant tumors in patients with sarcoidosis (P < 0.02) [18]. Malignant lymphoma and lung cancer were shown to occur 11 times and 3 times more frequently, respectively, than expected. The authors speculated that immunologic deficiencies related to sarcoidosis may predispose patients to malignancy. More recently, several prospective cohort studies have investigated the sarcoidosis – malignancy link. In a large cohort study, linking hospitalized sarcoidosis patients from a Swedish hospital registry with a national cancer registry, Ji et al reported a 40 percent overall excess occurrence of malignancy among sarcoidosis patients (overall standardized incidence ratio 1.40), attributable largely to squamous cell carcinoma, non-Hodgkin lymphoma, and leukemia. Cancer risk was especially pronounced within the first year post-hospitalization (SIR 1.18 beyond the first year of follow-up) [19]. Linkage analysis, by Reich et al. on patients cross-matched between a large health maintenance organization tumor registry and sarcoidosis registry, suggests that sarcoidosis and malignancy may be related pathogenetically (etiologically) in about 25 percent of the cases in which both occur concomitantly. The authors proposed that the link between sarcoidosis and malignancy might be related to a generalized cell mediated response to tumor antigens which are dispersed in the setting of an underlying malignancy [20].
Given that cutaneous manifestations are present in about one-quarter of patients with sarcoidosis and, furthermore, that sarcoidal skin lesions may occur in isolation without internal organ involvement, further investigation to examine a possible relationship between cutaneous sarcoidosis and malignancy is warranted. We have sought to characterize the presence and clinical features of cutaneous sarcoidosis in relationship with cancer and with other systemic manifestations of this disease in patients who display the co-existence of sarcoidosis with cancer.
Materials and methods
We reviewed the relevant English medical literature published on PubMed using the search terms “sarcoidosis and cancer” and “sarcoidosis and neoplasia” in addition to the other search terms “lymphoma,” “Hodgkin’s,” “melanoma,” “adenocarcinoma,” and “sarcoma.” This literature search revealed 100 well-documented cases of sarcoidosis associated with cancer.
Our case series comprised 10 cases of sarcoidosis associated with diverse malignancies, seen in the Dermatology Clinic at Georgetown University in the past 20 years. (Tables 1 to 4, bottom). A total of 110 cases of cutaneous and non-cutaneous sarcoidosis associated with malignancy were found and were included in the present analysis.
Data was collected after ethical approval from the Institutional Review Board, and consisted of patient’s age, gender, ethnicity, age and time of onset of sarcoidosis and malignancy, as well as histological documentation of sarcoidosis and the neoplasm(s).
Results
A review of the relevant medical literature identifies that both cutaneous sarcoidosis (Table 1 and Table 2) and extracutaneous sarcoidosis (Table 3 and Table 4) have been reported in patients who have also been diagnosed with malignancy. These associated neoplasms may occur either before, after, or concurrent with the diagnosis of sarcoidosis.
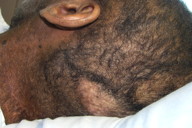 | 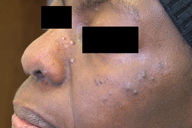 |
| Figure 1a | Figure 1b |
|---|---|
| Figure 1. Clinical aspects of cutaneous sarcoidosis in patients with associated cancer. Exemplified are cases of prostate cancer (1a) and breast cancer (1b). | |
We identified 100 cases reported in the literature of sarcoidosis associated with malignancy (Tables 1 through 4). This includes 58 cases of cutaneous sarcoidosis (58%); sarcoidosis preceded malignancy in 43 of these cases (74%). Furthermore, a total of 42 cases of non-cutaneous sarcoidosis associated with malignancy were retrieved. Among these, sarcoidosis preceded cancer in 33 cases (79%). Overall, with and without the presence of skin involvement, sarcoidosis was diagnosed before the detection of an associated neoplasm in a majority (76%) of cases, which is consistent with previous findings in the literature [18, 21-107]. Among these 100 cases of cutaneous (n=58) and non-cutaneous (n=42) sarcoidosis, hematologic malignancies accounted for 73 percent of cases, and sarcoidosis preceded the detection of neoplasia in a majority (76%) of cases.
We also present a novel series of 10 patients with sarcoidosis and associated malignancy who were evaluated in the past 20 years (Tables 1 through 4, bottom). Among these, 9 out of 10 were diagnosed with sarcoidosis prior to malignancy; the time between the two diagnoses ranged from 1 to 20 years. Four of these nine cases (44.4%) were cutaneous sarcoidosis (cutaneous sarcoidisis represented 40 percent of all sarcoidosis cases regardless of the timing of development of the neoplsia). Only one patient in our series developed non-cutaneous sarcoidosis and this occurred after the diagnosis of a colon cancer. In our series, all cases of cutaneous sarcoidosis preceding neoplasia presented solid tumors, including breast (n=4 tumors), prostate cancer, colon cancer, kidney cancer, and squamous cell carcinoma of the skin (n=1 of each type). The number of systemic malignancies for each patient with cutaneous sarcoidosis varied from 1 to 3, with an average of 2.0 cancers per patient. Two patients with cutaneous sarcoidosis developed three tumors each. Among the 6 patients in our series with cancers and non-cutaneous sarcoidosis, the types of neoplasias encountered were renal cancer (n=1), mycosis fungoides (n=1), diffuse large B-cell lymphoma (n=1), colon cancer (n=1), and ADK of parotid (n=2). The number of sarcoidosis cases seen during the time period when our 10 sarcoidosis-neoplasia cases occurred was approximately 300, corresponding to a significant 3.3 percent incidence of neoplasia in the overall sarcoidosis patient population.
Overall, among 110 total cases analyzed in this paper, cutaneous sarcoidosis was confirmed in 56.4 percent of cases, a figure exceeding expected rates of cutaneous involvement (20-25%) in the general sarcoidosis population. The majority of patients were young, with 80.4 percent of cases being 60 years of age or less.
Discussion
Physicians’ ability to recognize the link between sarcoidosis and malignancy is important. Dermatologists, in particular, may play a critical role in the recognition of cancer in these patients because many cases of sarcoidosis in association with cancer manifest initially, or even solely, with sarcoidal skin findings that may herald the development of cancer by several years (Tables 1-4). Therefore, patients who present with granulomatous skin lesions should be considered for screening for systemic sarcoidosis, as well as recommended for close follow-up (including age-appropriate cancer screening) to detect the emergence of associated disorders. Patients with sarcoidosis that is worsening, demonstrating new or evolving skin lesions, appear to be at the highest risk for malignancy. Although specific guidelines and risk-assessment tools for an early detection of malignancy have not yet been developed and validated, an increased physician awareness regarding the possible development and presence of systemic symptoms of cancer may contribute to better patient outcomes. Sarcoidosis skin findings provide both visible clues to the diagnosis and accessible tissue for histological examination.
Hematological malignancies remain most strongly linked with sarcoidosis compared to solid tumors. Since Brincker’s initial investigation, several observational and epidemiological studies in the past thirty years have described associations between sarcoidosis and multiple hematological disorders, including: Hodgkin lymphoma [99, 108], non-Hodgkin lymphoma (NHL) [33, 109], Mycosis Fungoides [21, 24], acute myeloid leukemia (AML) [10, 59, 88], chronic myelomonocytic leukemia (CMML) [51], hairy cell leukemia (HCL) [49], and multiple myeloma [111]. Among these, the link between sarcoidosis and lymphoma is best supported. The term sarcoidosis-lymphoma syndrome emerged in 1986 [109] to initially describe the development of lymphoma 1-2 years following a diagnosis of sarcoidosis. Patients associated with the sarcoidosis-lymphoma syndrome typically have a history of chronically active disease. It is estimated that patients with chronic sarcoidosis have a 5.5 times higher risk of developing a lymphoproliferative disorder [109].
The relationship between sarcoidosis and solid tumors is less extensively reported. The strongest association is described with adenocarcinoma of the lung [44], although other cancers have also been reported, including: small cell lung cancer [112], testicular cancer [113], carcinoid tumors [114], gastric adenocarcinoma [48], bladder cancer [115], adenocarcinoma of the colon [116], melanoma [104], Kaposi sarcoma [117], adenocarcinoma of the prostate [57], esophageal adenocarcinoma [38], hepatocellular carcinoma [100], papillary carcinoma of the thyroid [60], malignant lymphoplasmacytoid lymphoma (immunocytoma), squamous cell carcinoma of the uterine cervix [73], squamous cell carcinoma of the gingiva [102], and sweat gland carcinoma [22]. Whereas the individual incidences of lung cancer and sarcoidosis are relatively prevalent, their occurrence in the same patient is very rare. In Brincker’s initial report [18], a threefold excess of lung cancers was noted in patients with sarcoidosis. Several case reports describe the coexistence of these two disorders [93]. In most instances, sarcoidosis preceded lung cancer by several years. Several hypotheses have emerged to explain this association. It is possible that the two diseases exist purely by chance. One premise is that the immune dysfunction related to sarcoidosis predisposes to later development of lung cancer. Alternatively, lung cancers in the setting of sarcoidosis may represent scar cancers, which develop in pre-existing fibrous tissue of sarcoid nodules [118]. Immune suppressive and mutagenic effects of the therapies used in sarcoidosis are other important factors to be considered for the development of secondary malignancies.
Whereas the association between sarcoidosis and malignancy is well documented, the mechanisms explaining these observations remain undetermined. Several theories related to immune dysregulation, impairment of cellular immunity, and other sequelae of chronic inflammation have been proposed (discussed in Dermatology Online Journal current issue, January 2011, Volume 17, Number 1, Commentary “On the missing link between inflammation and cancer”). The strength of the association is further enhanced by the development of neoplasias in relatively young patients, which is demonstrated in our analysis; less than 20 percent of patients were above the age of 60. Future therapies targeting inflammation will need to be tailored against the cellular pathways that also favor the development of cancers.
Because many instances of malignancies associated with sarcoidois are in fact not likely to be reported, it is probable that the risk of cancer in this setting remains under-estimated. Our results suggest that cutaneous sarcoidosis is present in a majority of cases of sarcoidosis associated with cancer (56.4%), more than the frequency of cutaneous involvement in the general population of sarcoidosis patients (20-25%). These findings support an etiological role of cutaneously located sarcoidosis, or, alternatively, may indicate that the skin disease represents a marker for a particular sarcoidosis phenotype associated with internal malignancy.
References
1. Zax RH, Callen JP. Granulomatous Reactions. In: Sams WM Jr., Lynch PJ, eds. Principles and Practices of Dermatology. 2nd ed. New York, NY: Churchill Livingstone; 1996:629-632.2. Mañá J, Marcoval J, Graells J, Salazar A, Peyrí J, Pujol R. Cutaneous involvement in sarcoidosis. Relationship to systemic disease. Arch Dermatol. 1997;133:882-8. Review. [PubMed]
3. Anonymous. Statement on sarcoidosis. Joint Statement of the American Thoracic Society (ATS), the European Respiratory Society (ERS) and the World Association of Sarcoidosis and Other Granulomatous Disorders (WASOG) adopted by the ATS Board of Directors and by the ERS Executive Committee, February 1999. Am J Respir Crit Care Med. 1999;160:736-55. Review. [PubMed]
4. Hanno R, Callen JP. Sarcoidosis: a disorder with prominent cutaneous features and their interrelationship with systemic disease. Med Clin North Am. 1980;64:847-66. [PubMed]
5. Hanno R, Needelman A, Eiferman RA, Callen JP. Cutaneous sarcoidal granulomas and the development of systemic sarcoidosis. Arch Dermatol. 1981;117:203-7. [PubMed]
6. Veien NK, Stahl D, Brodthagen H. Cutaneous sarcoidosis in Caucasians. J Am Acad Dermatol. 1987;16:534-40. [PubMed]
7. Rodionov AN, Samtsov AV. Correlation between cutaneous sarcoidosis and systemic sarcoidosis. Acta Derm Venereol. 1992;72:153-4. [PubMed]
8. Newman LS, Rose CS, Maier LA. Sarcoidosis. N Engl J Med. 1997;336:1224-34. [PubMed]
9. Sharma OP. Sarcoidosis of the Skin. Fitzpatrick’s Dermatology in General Medicine, 5th edn. St. Louis: McGraw-Hill, 1999:2099-2106.
10. Yanardağ H, Pamuk ON, Karayel T. Cutaneous involvement in sarcoidosis: analysis of the features in 170 patients. Respir Med. 2003;97:978-82. [PubMed]
11. Cancrini C, Angelini F, Colavita M, Cortis E, Chini L, Mammone F, Rossi P, De Sanctis R. Erythema nodosum: a presenting sign of early onset sarcoidosis. Clin Exp Rheumatol. 1998;16:337-9. [PubMed]
12. Scadding JG Mitchell DN Sarcoidosis of the skin. In: Scadding JG, Mitchell DN, eds. Sarcoidosis. London, England: Chapman&Hall Medical; p. 181–206. 1985.
13. James DG, Neville E, Siltzbach LE. A worldwide review of sarcoidosis. Ann N Y Acad Sci. 1976;278:321-34. [PubMed]
14. Kerdel FA, Moschella SL. Sarcoidosis. An updated review. J Am Acad Dermatol. 1984;11:1-19. [PubMed]
15. James DG. Dermatological aspects of sarcoidosis. Q J Med. 1959;28:108-24. [PubMed]
16. Elgart ML. Cutaneous sarcoidosis: definitions and types of lesions. Clin Dermatol. 1986;4:35-45. [PubMed]
17. Kashyap S, Kumar M, Thami GP, Saini V. Umbilicated papular sarcoidosis. Clin Exp Dermatol. 1996;21:395-6. [PubMed]
18. Brincker H, Wilbek E. The incidence of malignant tumours in patients with respiratory sarcoidosis. Br J Cancer. 1974;29:247-51. [PubMed]
19. Ji J, Shu X, Li X, Sundquist K, Sundquist J, Hemminki K. Cancer risk in hospitalized sarcoidosis patients: a follow-up study in Sweden. Ann Oncol. 2009;20:1121-6. Epub 2009 Feb 11. [PubMed]
20. Reich JM, Mullooly JP, Johnson RE. Linkage analysis of malignancy-associated sarcoidosis. Chest. 1995;107:605-13. [PubMed]
21. Atwood WG, Miller RC, Nelson CT. Sarcoidosis and the malignant lymphoreticular diseases. Arch Dermatol. 1966;94:144-51. [PubMed]
22. Asvesti C, Hadziyannis SJ, Papadopoulos S, Giakoumoglou T, Hadziolou E, Trichia E, Zographakis I. Sweat gland carcinoma in a patient with sarcoidosis and primary biliary cirrhosis. Dermatology. 1994;189:198-202. [PubMed]
23. Banno S, Takada K, Wakita A, Iwaki O, Nitta M, Mitomo Y, Yamamoto M. Non-Hodgkin's lymphoma in a patient with sarcoidosis (the sarcoidosis-lymphoma syndrome. Rinsho Ketsueki. 1992 Aug;33(8):1066-70. [PubMed]
24. Bessis D, Sotto A, Farcet JP, Barnéon G, Guilhou JJ. Granulomatous mycosis fungoides presenting as sarcoidosis. Dermatology. 1996;193:330-2. [PubMed]
25. Brechtel B, Haas N, Henz BM, Kolde G. Allopurinol: a therapeutic alternative for disseminated cutaneous sarcoidosis. Br J Dermatol. 1996 Aug;135(2):307-9. [PubMed]
26. Cantwell AR Jr. Variably acid-fast bacteria in a rare case of coexistent malignant lymphoma and cutaneous sarcoid-like granulomas. Int J Dermatol. 1982 Mar;21(2):99-106. PubMed [PubMed] [PubMed]
27. Caras WE, Dillard T, Baker T, Pluss J. Coexistence of sarcoidosis and malignancy. South Med J. 2003 Sep;96(9):918-22. [PubMed]
28. Chao SC, Yan JJ, Lee JY. Cutaneous sarcoidosis among Taiwanese. J Formos Med Assoc. 2000 Apr;99(4):317-23. [PubMed]
29. Collet E, Boulitrop-Morvan C, Dalac S, Camus P, Lambert D. Association of sarcoidosis and Waldenström's macroglobulinemia. Ann Dermatol Venereol. 1992;119(12):971-4. [PubMed]
30. Corda L, Benerecetti D, Ungari M, Facchetti F, Radaeli E. Kaposi's disease and sarcoidosis. Eur Respir J. 1996 Feb;9(2):383-5. [PubMed]
31. Diette KM, Caro WA, Roenigk HH Jr. Malignant lymphoma presenting with cutaneous granulomas. J Am Acad Dermatol. 1984 May;10(5 Pt 2):896-902. [PubMed]
32. Falchero L, Nesme P, Perol M, Guérin JC. Spontaneous resolution of multiorgan sarcoidosis associated with malignant lymphoma. Rev Mal Respir. 1998 Jun;15(3):287-9. [PubMed]
33. Harada H, Shijubo N, Katoh S, Satoh M, Abe S. Malignant T cell lymphoma associated with sarcoidosis. Sarcoidosis. 1994;11:132-4. [PubMed]
34. Hermann C, Andersen E, Videbaek A. Acute myeloblastic leukaemia in sarcoidosis treated with methotrexate. Scand J Haematol. 1980 Mar;24(3):234-6. [PubMed]
35. Karakantza M, Matutes E, MacLennan K, O'Connor NT, Srivastava PC, Catovsky D. Association between sarcoidosis and lymphoma revisited. J Clin Pathol. 1996 Mar;49(3):208-12. [PubMed]
36. Katz KA. Disseminated cutaneous granulomatous eruption occurring in the setting of myelodysplasia. Dermatol Online J. 2003 Oct;9(4):22. [PubMed]
37. Keohane SG, Savin JA, Tidman MJ, Anderson JA, Carey FA. The sarcoidosis-lymphoma syndrome: acceleration of the cutaneous sarcoidosis during chemotherapy of the lymphoma. Acta Derm Venereol. 1996 May;76(3):251-3. [PubMed]
38. Maekawa Y, Nogami R. A case of progressive systemic sclerosis associated with sarcoidosis and esophageal adenocarcinoma. J Dermatol. 1993;20:45-8. [PubMed]
39. Maillard H, Croué A, François S, Verret JL. Granulomatous mycosis fungoides histologically simulating cutaneous sarcoidosis. Ann Dermatol Venereol. 1998 Dec;125(12):912-5. [PubMed]
40. Mainguene C, Picard O, Audouin J, Le Tourneau A, Jagueux M, Diebold J. An unusual case of mycosis fungoides presenting as sarcoidosis or granulomatous mycosis fungoides. Am J Clin Pathol. 1993 Jan;99(1):82-6. [PubMed]
41. McFarland JP, Kauh YC, Luscombe HA. Sarcoidosis associated with mycosis fungoides. Arch Dermatol. 1978 Jun;114(6):912-5. [PubMed]
42. Morii S, Oka K, Naoi Y, Kotsuji T, Nihei T, Nagayama R, Kashimura K, Kameta S, Yatabe Y, Mori N. Coexisting primary early gastric plasmacytoma and sarcoidosis with hypercalcaemia. Virchows Arch. 1998 May;432(5):473-6. [PubMed]
43. Myerson RM, Soroush A, Skerrett PV. Hepatocellular carcinoma, positive Australia (hepatitis-associated) antigen and sarcoidosis. Am J Dig Dis. 1971 Sep;16(9):857-62. [PubMed]
44. Noone PG, O'Briain DS, Luke D, Clancy L. Adenocarcinoma of the lung in association with chronic sarcoidosis. Ir Med J. 1993;86:27-8. [PubMed]
45. Okada M, Tanaka M, Horiuchi N, Kato T, Tagami H. Coexistence of xeroderma pigmentosum with sarcoidosis and adenocarcinoma of the digestive organs. Acta Derm Venereol. 2000 Sep-Oct;80(5):376-7. [PubMed]
46. Oliwiecki S, Kotecha B, Kingston T, Rothera MP. Sarcoidosis-lymphoma syndrome. J R Soc Med. 1992 Mar;85(3):176-7. [PubMed]
47. Raben AC, Bogdanovich NK, Golochevskaya VS. A case of transformation of sarcoidosis into reticulosarcomatosis. Probl Gematol Pereliv Krovi. 1961;6:763-9. [PubMed]
48. Roth D, West B, Madison J, Cooper D. Gastric carcinoma in a patient with sarcoidosis of the gastrointestinal tract. Am J Gastroenterol. 1994;89:1589-91. [PubMed]
49. Schiller G, Said J, Pal S. Hairy cell leukemia and sarcoidosis: a case report and review of the literature. Leukemia. 2003;17:2057-9. [PubMed]
50. Sherertz EF, Westwick TJ, Flowers FP. Sarcoidal reaction to lymphoma presenting as granulomatous rosacea. Arch Dermatol. 1986 Nov;122(11):1303-5. [PubMed]
51. Serratrice J, Granel B, Swiader L, Disdier P, Weiller-Merli C, Lepidi H, de Roux-Serratrice C, Andrac-Meyer L, Weiller PJ. Sarcoidosis preceding chronic myelomonocytic leukemia. Report of two cases. Dermatology. 2002;205:409-12. [PubMed]
52. Simsek S, van Leuven F, Bronsveld W, Ooms GH, Groeneveld AB, de Graaff CS. Unusual association of Hodgkin's disease and sarcoidosis. Neth J Med. 2002;60:438-40. [PubMed]
53. Sorrells T, Pratt L, Newton J, Graham S, Ryan M. Spontaneous regression of granulomatous mycosis fungoides in an HIV positive patient. J Am Acad Dermatol. 1997 Nov;37(5 Pt 2):876-80. [PubMed]
54. Suarez-Vilela D, Izquierdo-Garcia FM. Angioimmunoblastic lymphadenopathy-like T-cell lymphoma: cutaneous clinical onset with prominent granulomatous reaction. Am J Surg Pathol. 2003 May;27(5):699-700. [PubMed]
55. Takada K, Ina Y, Noda M, Sato T, Yamamoto M, Morishita M. The clinical course and prognosis of patients with severe, moderate or mild sarcoidosis. J Clin Epidemiol. 1993 Apr;46(4):359-66. PubMed [PubMed]
56. Terao S, Machida K, Shiomi H, Yamasaki Y, Iwata K. A case of sarcoidosis presenting with hoarseness due to false vocal cord tumor. Nihon Kyobu Shikkan Gakkai Zasshi. 1984 Jun;22(6):514-7. [PubMed]
57. Todd RF 3rd, Garnick MB. Prostatic adenocarcinoma, sarcoidosis and hypercalcemia: an unusual association. J Urol. 1980 Jan;123:133-4. [PubMed]
58. Ueda T, Tsutani H. Primary extranodal malignant lymphoma. Nippon Rinsho. 2000 Mar;58(3):673-6. [PubMed ]
59. Vestey JP, Turner M, Biddlestone L, McLaren K, Goulden N, Hunter JA. Disseminated cutaneous granulomatous eruptions associated with myelodysplastic syndrome and acute myeloid leukaemia. Clin Exp Dermatol. 1993;18:559-63. [PubMed]
60. Zimmermann-Belsing T, Christensen L, Hansen HS, Kirkegaard J, Blichert-Toft M, Feldt-Rasmussen U. A case of sarcoidosis and sarcoid granuloma, papillary carcinoma, and Graves' disease in the thyroid gland. Thyroid. 2000;10:275-8. [PubMed]
61. Farrell AM, Henry K, Woodrow D, Francis N, Newlands ES, Mitchell DN, Cream JJ. Cutaneous granulomas associated with high-grade T-cell non-Hodgkin's lymphoma. Br J Dermatol. 1999 Jan;140(1):145-9. PubMed [PubMed]
62. Kornacker M, Kraemer A, Leo E, Ho AD. Occurrence of sarcoidosis subsequent to chemotherapy for non-Hodgkin's lymphoma: report of two cases. Ann Hematol. 2002 Feb;81(2):103-5. [PubMed]
63. Kikawada M, Ichinose Y, Kunisawa A, Yanagisawa N, Minemura K, Kasuga I, Yonemaru M, Kawanishi K, Takasaki M, Toyama K. Sarcoidosis induced by interferon therapy for chronic myelogenous leukaemia. Respirology. 1998 Mar;3(1):41-4. [PubMed]
64. Mauduit G, Souteyrand P, Cambazard F, Faure M, Hermier C, Thivolet J. Non-epidermotropic malignant cutaneous lymphoma and sarcoidosis. Ann Dermatol Venereol. 1983;110(1):59-62. French. PubMed [PubMed]
65. Marzano AV, Gasparini G, Caputo R, Alessi E. Subcutaneous sarcoidosis following hypophysectomy for pituitary microadenoma inducing Cushing's disease. Int J Dermatol. 1998 Oct;37(10):798. [PubMed]
66. Pujol RM, Matías-Guiu X, Planagumà M, de Moragas JM. Chronic lymphocytic leukemia and cutaneous granulomas at sites of herpes zoster scars. Int J Dermatol. 1990 Nov;29(9):652-4. [PubMed]
67. Robert C, Schoenlaub P, Avril MF, Lok C, Grosshans E, Valeyre D, Bourgeois C, Pinquier L, Dubertret L, Guillaume JC. Malignant melanoma and granulomatosis. Br J Dermatol. 1997 Nov;137(5):787-92. [PubMed]
68. Schmuth M, Prior C, Illersperger B, Topar G, Fritsch P, Sepp N. Systemic sarcoidosis and cutaneous lymphoma: is the association fortuitous? Br J Dermatol. 1999 May;140(5):952-5. [PubMed]
69. Takenaka K, Yanase T, Takayanagi R, Haji M, Ikuyama S, Nawata H. Cushing's disease preceding sarcoidosis. Intern Med. 1995 Jun;34(6):580-3. [PubMed]
70. Yavorkovsky LL, Carrum G, Bruce S, McCarthy PL Jr. Cutaneous sarcoidosis in a patient with Philadelphia-positive chronic myelogenous leukemia treated with interferon-alpha. Am J Hematol. 1998 May;58(1):80-1. [PubMed]
71. Weltfriend S, Harth Y, Katz I. Subcutaneous sarcoidosis in a patient with malignant carcinoid tumor of the colon. J Am Acad Dermatol. 1989 Mar;20(3):507-8. [PubMed]
72. de Hemricourt E, De Boeck K, Hilte F, Abib A, Kockx M, Vandevivere J, De Bock R. Sarcoidosis and sarcoid-like reaction following Hodgkin's disease. Report of two cases. Mol Imaging Biol. 2003 Jan-Feb;5(1):15-9. [PubMed]
73. Alliot C, Barrios M, Desplechain C. Multisystem sarcoidosis and carcinoma of the uterine cervix: an unusual association. Int J Gynecol Cancer. 2001;11:323-5. [PubMed]
74. Brandt L, Emanuelsson H, Mitelman F, Stenstam M, Söderström N. Pronounced deficiency in T-cells and lymphocyte chromosomal aberrations in a patient with sarcoidosis, myelofibrosis and acute leukaemia following thorotrast angiography. Acta Med Scand. 1977;201(5):487-9. [PubMed]
75. Brennan NJ, Fennelly JJ, Towers RP, FitzGerald MX. Sarcoidosis and lymphoma in the same patient. Postgrad Med J. 1983 Sep;59(695):581-5. [PubMed]
76. Buckle RM. Reticulosarcoma complicating sarcoidosis. Tubercle. 1960 Jun;41:213-5. [PubMed]
77. Caras WE, Dillard T, Baker T, Pluss J. Coexistence of sarcoidosis and malignancy. South Med J. 2003 Sep;96(9):918-22. [PubMed]
78. de la Figuera M, Biosca M, García-Bragado F, Villar M, Magriñá C. Spontaneous regression of bilateral hilar lymphadenopathy in renal cell carcinoma. Eur J Respir Dis. 1985 Aug;67(2):133-5. [PubMed]
79. Echigo T, Saito A, Takehara K, Takata M, Hatta N. Coexistence of micrometastatic melanoma cells and sarcoid granulomas in all regional lymph nodes in a patient with acral melanoma. Clin Exp Dermatol. 2003 Jul;28(4):375-6. [PubMed]
80. Falcão RP, Voltarelli JC, Simões BP, Pestana DN, Zago MA, Figueiredo MS. Malignant T gamma/delta lymphoproliferative disease with natural killer lytic activity. Am J Hematol. 1992 Oct;41(2):128-31. [PubMed]
81. Finke R, Lydtin H, Prechtel K. Sarcoidosis and immunocytoma. Am J Med. 1986 May;80:939-42. [PubMed]
82. Foon KA, Filderman A, Gale RP. Histiocytic lymphoma following resolution of sarcoidosis. Histiocytic lymphoma following resolution of sarcoidosis. Med Pediatr Oncol. 1981;9(4):325-31. [PubMed]
83. Fukuda T, Sato K, Tachikawa S, Ohnuki K, Ohtani H, Suzuki T. Mucosa-associated lymphoid tissue lymphoma coexisting with epithelioid granulomas in the stomach of a patient with systemic sarcoidosis. Pathol Int. 1997 Dec;47(12):870-5. [PubMed]
84. Goldfarb BL, Cohen SS. Coexistent disseminated sarcoidosis and Hodgkin's disease. JAMA. 1970 Mar 2;211(9):1525-8. [PubMed]
85. Goldberg DJ, Robins P. Subungual squamous cell carcinoma treated by Mohs surgery in a patient with sarcoidosis. J Dermatol Surg Oncol. 1986 Sep;12(9):972-4. [PubMed]
86. Hatake K, Aoyagi N, Takaku F. Acute myelomonocytic leukemia following remission of sarcoidosis. Oncology. 1983;40(3):200-1. [PubMed]
87. Masuda R, Toyoshima H, Bandou T, Isoyama T, Matsui Y, Takemura T. Malignant lymphoma of the stomach associated with systemic sarcoidosis. Cancer. 1992 Dec 1;70(11):2592-6. [PubMed]
88. Murphy EA, Murphy JA, Jackson R, Sturrock RD. Sarcoidosis and acute myeloid leukaemia. J R Soc Med. 1991;84:368-9. [PubMed]
89. Panelatti G, Plumelle Y, Arfi S, Pascaline N, Caplanne D, Jean-Baptiste G. Sarcoidosis and leukemia/T-cell lymphoma associated with HTLV-1 virus infection in adults (apropos of a case). Rev Med Interne. 1992 Jul-Aug;13(4):299-301. [PubMed]
90. Pollock JL, Catalano E. Metastatic ductal carcinoma of the parotid gland in a patient with sarcoidosis. Arch Dermatol. 1979 Sep;115(9):1098-9. [PubMed]
91. Ponticelli P, Arganini L, Cionini L. Hodgkin's disease associated with sarcoidosis: case report. Tumori. 1981 Feb 28;67(1):45-51. [PubMed]
92. Romero J, Ramón y Cajal S, Vargas JA, Pérez-Maestu R, Salas C, Regueiro CA, Aragon G. Association of sarcoidosis, low-grade B-lymphoma and epidermoid carcinoma. Postgrad Med J. 1997 Sep;73(863):580-2. [PubMed]
93. Sarkar TK. Anaplastic carcinoma of the lung and sarcoidosis. Br J Clin Pract. 1970;24:297-9. [PubMed]
94. Sen F, Mann KP, Medeiros LJ. Multiple myeloma in association with sarcoidosis. Arch Pathol Lab Med. 2002 Mar;126(3):365-8. [PubMed]
95. Silver HM, Nachnani G, Breslow A. Lymphosarcoma and sarcoidosis. Simpson JR. Sarcoidosis with erythrodermia and ulceration. Br J Dermatol. 1963;75:193-8. [PubMed]
96. Spitzer T, Crum E, Schacter L, Abboud S. Sarcoidosis, Hodgkin's disease, and autoimmune hemolytic anemia. Am J Med Sci. 1986 Mar;291(3):190-3. [PubMed]
97. Stoker TA, Ellis H. Hodgkin's disease with sarcoid features. Proc R Soc Med. 1971 Jun;64(6):661. [PubMed]
98. Tamura A, Hashimoto K, Tanoue M, Murota N, Ohdama S, Akagawa S, Matsubara O, Kamiyama R, Tanaka T, Mitsunaga K. A case of so-called Lennert's lymphoma, suspected of sarcoidosis due to uveitis occurring first and elevation of serum angiotensin-converting enzyme (ACE). Nihon Kyobu Shikkan Gakkai Zasshi. 1984 Nov;22(11):1033-9. [PubMed]
99. Trier H, Christensen M. Hodgkin's disease in sarcoidosis. A case report and a review of the literature. Acta Med Scand. 1985;218:137-40. [PubMed]
100. Wong VS, Adab N, Youngs GR, Sturgess R. Hepatic sarcoidosis complicated by hepatocellular carcinoma. Eur J Gastroenterol Hepatol. 1999;11:353-5. [PubMed]
101. Cheng DS, Kitahara M, Logan KH. Chronic granulocytic leukemia: long-term remission in a patient with familial sarcoidosis. South Med J. 1979;72:645-7. [PubMed]
102. Hanibuchi M, Matsumori Y, Nishioka Y, Sone S. A case of sarcoidosis accompanying squamous cell carcinoma in the mandibular gingiva. J Med Invest. 2005;52:118-21. [PubMed]
103. Haran MZ, Feldberg E, Berrebi A. Lymphoma masking sarcoidosis. Leuk Lymphoma. 2002 Aug;43(8):1709-10. PubMed [PubMed]
104. Mackenzie, Retsas S. Malignant melanoma co-existing with sarcoidosis: implications for prognosis and management. Melanoma Res. 1996 Feb;6(1):71-2. [PubMed]
105. Nakamura Y, Kurihara N, Sato A, Nakamura M, Koyama K, Suzuki H, Saito H, Ishibashi T, Takahashi S. Muscle sarcoidosis following malignant lymphoma: diagnosis by MR imaging. Skeletal Radiol. 2002 Dec;31(12):702-5. Epub 2002 Jun 26. PubMed [PubMed]
106. Rubinstein I, Baum GL, Yellin A, Herczeg E. Sarcoidosis: a cause of bilateral hilar lymphadenopathy after excision of malignant melanoma of the arm. South Med J. 1985 Sep;78(9):1139-40. PubMed [PubMed]
107. van den Berg, Fickers M, Theunissen P, van Noord JA. Pulmonary sarcoid-like granulomata in a patient treated for extrapulmonary Hodgkin's disease. Respiration. 1997;64(1):114-7. PubMed [PubMed]
108. Simsek S, van Leuven F, Bronsveld W, Ooms GH, Groeneveld AB, de Graaff CS. Unusual association of Hodgkin's disease and sarcoidosis. Neth J Med. 2002 Dec;60(11):438-40. [PubMed]
109. Brincker H. The sarcoidosis-lymphoma syndrome. Br J Cancer. 1986;54:467-73. [PubMed]
110. Reich JM. Acute myeloblastic leukemia and sarcoidosis. Implications for pathogenesis. Cancer. 1985;55:366-9. [PubMed]
111. Uchiumi H, Murakami H, Matsushima T, Tamura J, Sawamura M, Naruse T, Tsuchiya J. Does sarcoidosis induce multiple myeloma? Am J Hematol. 1993;44:220. [PubMed]
112. Conejo-Mir JS, Casals M, Garciandia C, Artola JL, Quintana del Olmo J, Rivera Hueto F, Navarrete M. Cutaneous sarcoid granulomas with oat cell carcinoma of the lung. Dermatology. 1995;191:59-61. [PubMed]
113. Rayson D, Burch PA, Richardson RL. Sarcoidosis and testicular carcinoma. Cancer. 1998;83:337-43. [PubMed]
114. Levy NT, Rubin J, DeRemee RA, Aughenbaugh GL, Unni KK, Kahn MJ. Carcinoid tumors and sarcoidosis--does a link exist? Mayo Clin Proc. 1997;72:112-6. [PubMed]
115. Suzuki M, Ohya K, Nagamatsu H, Mizuo T. Sarcoidosis in a young woman with bladder carcinoma. J Urol. 1998;160:123. [PubMed]
116. Mohamadnejad M, Babai M, Bidari A, Malekzadeh R, Tavangar SM. Adenocarcinoma of the colon associated with sarcoidosis. MedGenMed. 2003;5:6. [PubMed]
117. Dessoukey MW, Dayem HA, Omer MF. Kaposi's sarcoma and sarcoidosis coexisting in lesions of HIV-seronegative patient. Int J Dermatol. 1996;35:824-6. [PubMed]
118. Brincker H. Sarcoidosis and malignancy. Chest. 1995;108:1472-4. [PubMed]
© 2011 Dermatology Online Journal

