Eccrine angiomatous hamartoma: A rare multifocal variant with features suggesting trauma
Published Web Location
https://doi.org/10.5070/D33fd7h8bsMain Content
Eccrine angiomatous hamartoma: A rare multifocal variant with features suggesting trauma
Veena Naik MD1, Nebojsa Arsenovic MD2, Malcolm Reed MD2
Dermatology Online Journal 15 (9): 6
1. SpR. Path2. Spec. Path
Department of Cellular Pathology, Pathlinks Pathology Services, Lincoln County Hospital, Lincoln, United Kingdom
Abstract
Eccrine angiomatous hamartoma (EAH) is a rare, benign cutaneous tumor characterized by proliferation of the eccrine gland elements closely associated with capillary angiomatosis and proliferation of other dermal elements, such as adipose tissue, hair and epidermis. Patients usually present with a solitary nodule on the extremities appearing at birth or during the prepubertal years. However multifocal lesions or late onset of this condition may occur. Eccrine angiomatous hamartoma is usually sporadic, but one familial case of the multifocal variant has been reported. The clinical presentation ranges from a simple angiomatous nodule to erythematous – purpuric plaques. Eccrine angiomatous hamartoma is generally asymptomatic but may occasionally be associated with pain and hyperhidrosis. We report a rare case of the multifocal variant of EAH in a 13-year-old girl, with histological features suggesting trauma. Clinically, this condition must be differentiated from other angiomatoses and a definitive diagnosis is based upon histology. Eccrine angiomatous hamartoma is a benign slowly growing lesion for which aggressive treatment is not indicated. Simple excision is reserved for painful or cosmetically disfiguring examples.
Case report
Clinical data
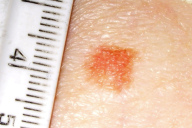 |
| Figure 1 |
|---|
| Figure 1. One of the abdominal skin lesions before biopsy |
A 13-year-old girl presented to our outpatient clinic with multifocal erythematous macular papules on the abdomen, back, and thighs, which had been present since birth and had enlarged with age. She had never had any medical consultation for her skin condition. There was significant medical history for mild learning disability. However, "a few months back," she had noticed a change on two abdominal lesions described as a "narrow, pale brown ring around, which was fading." Physical examination revealed very dry, older looking skin with multifocal angiomatous macules, 0.8 to 1.6 cm in diameter, symmetrically involving her trunk and extremities. There was no hyperhidrosis on the surface of the macules. Two lesions were tendermacules, 0.8 cm in diameter each, and 2 cm away from each other. They were red to maroon, relatively symmetrical and well circumscribed, and localized on the lower abdomen with evidence of mild belt pressure over them. However, no discrete change around the lesions was observed. One of the described lesions (Fig. 1) was biopsied with the clinical suspicion of "blue rubber bleb nevus."
Microscopic findings with immunoprofile
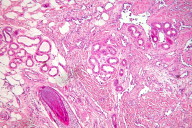
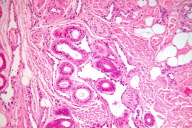
Histological examination revealed a poorly defined abnormality in the deep dermis, which was composed of increased number of secretory and ductal portions of eccrine glands intimately admixed with irregular, dilated, thin-walled and variably sized vascular elements and fat lobules (Figs. 2a & 2b). The vessels dissected collagen bundles. Intraluminally they had papillary projections lined by prominent endothelial cells with a hobnail appearance. In some parts of the lesion the hyalinized tips of the papillae appeared to float freely within the luminae (Fig. 2a). A lobule of the eccrine gland had been dissected by an irregular, dilated and thin-walled vascular channel showing an almost free floating glandular duct within its lumen (Fig. 2b).
Immunohistochemical studies showed that the eccrine elements were positive for carcinoembryonic antigen (CEA) (Fig. 3), whereas the endothelial vascular component displayed a strong positive reaction for CD34 (Fig. 4). These microscopic findings were consistent with the diagnosis of an eccrine angiomatous hamartoma with reactive vascular changes suggesting trauma.
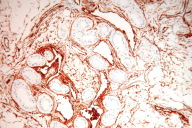 | 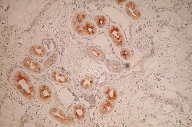 |
| Figure 3 | Figure 4 |
|---|---|
| Figure 3. The vascular component of EAH is highlighted by CD34 immunostain. Figure 4. The eccrine secretory and ductal structures of EAH are highlighted by CEA immunostain. | |
Discussion
Eccrine angiomatous hamartoma is a rare benign cutaneous hamartoma, composed of proliferating eccrine secretory coils and ducts intimately associated with capillary angiomatous channels and occasionally other minor elements, such as fatty lobules and hair [1]. First described by Lotzbeck in 1859 as an angiomatous appearing lesion on the cheek of a child, the term EAH was coined by Hymann and co-workers in 1968 [3]. Eccrine angiomatous hamartoma usually occurs as a solitary lesion, but cases with multiple lesions have been described [6, 12, 13, 14]. Classically it presents in childhood but there are reports of adult onset cases up to age 71 [11]. It may or may not be associated with hyperhidrosis and pain. The infiltration of small nerves may be responsible for this pain, and local increases in the temperature related to the angioma may produce the sweating [4, 5]. Histopathologically EAH is a relatively circumscribed/lobular, unencapsulated, dermal proliferation of mature appearing eccrine secretory and ductal structures, intimately associated with thin walled angiomatous channels, usually of a capillary nature but of variable size. The lesion usually has a lobular appearance, though rare ill-defined lesions have been described [9]. Eccrine angiomatous hamartoma has been seen to be associated with a number of other histological elements including adipose tissue, epidermis, mucin, apocrine glands, neural elements, and neurovascular glomic bodies [2, 5, 6, 16]. Immunohistochemical studies on EAH have been done demonstrating the presence of antigens commonly found in eccrine glands, such as carcinoembryonic antigen (CEA) and S-100.
However, the intensity of antigen binding to the eccrine elements was decreased [4, 5]. Other antigens demonstrated include CD34, CD44, factor VIII and Ulex europaeus antigens [4, 5, 7], as well as gross cystic disease fluid protein-15 in one case [15]. The etiology of EAH has not been delineated clearly. According to Zeller and Goldman it might be caused by abnormal induction of heterotypic dependency during organogenesis [8]. The clinical differential diagnosis of EAH includes eccrine nevus, blue rubber bleb nevus, and other angiomatoses such as tufted hemangioma [3, 9], macular telangiectatic mastocytosis, nevus flammeus, glomus tumor, and smooth muscle hamartoma [10].
In this paper we have described an EAH with a rather unusual vascular component, which in our opinion represents the consequence of local trauma. The above-mentioned vascular lesions in the differential diagnosis could be easily excluded by clinico-pathological correlations. However, because of the unusual histological features of the vascular component of our tumor, the differential diagnosis had to include other vascular proliferations, such as hobnail hemangioma, hyperkeratotic vascular stains, retiform hemangioendothelioma, benign lymphangioendothelioma, and Dabska tumor.
Hobnail hemangioma is an acquired, usually solitary lesion, which histologically shows a distinctive biphasic appearance. Within the papillary dermis are dilated, irregular, thin-walled vessels with some hyalinized fronds lined by endothelial cells with a hobnail appearance. Within a fibrotic deep dermis there are angulated, slit-shaped vascular spaces dissecting collagen [20]. No eccrine glandular elements are associated with the angiomatous channels. Hemosiderin, red blood cell extravasations and a sparse mononuclear lymphocytic infiltrate may be seen [17, 18].
Hyperkeratotic vascular stains appear at birth or shortly after and usually affect the lower limbs unilaterally. Histologically, it shows verrucous epidermal hyperplasia and ectatic, irregularly shaped vessels with walls of variable thickness [19]. The lesion involves the dermis and occasionally subcutis but is not associated with eccrine glands.
Benign lymphangioendotleioma [19] is typically a bruise like, solitary lesion appearing during adolescence. Histologically, within the papillary dermis there are delicate, horizontally arranged, cleft-like, thin-walled, endothelial-lined spaces becoming smaller at deeper levels. They may dissect preexisting vessels and adnexal structures of the dermis and have discrete papillary intraluminal fronds, but are not described as dilated, irregular, or intimately associated with eccrine glands as in EAH.
Retiform hemangioendothelioma is a distinctive variant of low-grade angiosarcoma of the skin usually described as a slowly growing solitary lesion on the extremities of adults.
Dabska tumor is a variant of low-grade angiosarcoma in children. The usually affected sites are skin and subcutis of the head, neck, and limbs. Microscopically, it is characterized by interconnecting vascular channels of different size and shape with intraluminal papillary plugs of atypical endothelial cells with a central hyalinized core [19].
In conclusion, EAH is a benign, slowly growing hamartomatous proliferation. Simple excision is only needed for cosmetic reasons or for intractable pain. The presented case was the multifocal variant, clinically suspected to be blue rubber bleb nevus based on the clinical appearance.
Histologically, however, it was proven to be EAH with a microscopically ill-defined growth pattern and some features suggesting previous trauma. Similar features of papillary fronds with hyalinized central cores lined by endothelial cells with a hobnail appearance, extending into the lumina, and apparently free intraluminal papillae, have been described in cases of intravascular papillary endothelial hyperplasia and hobnail hemangioma. Furthermore, in cases of hobnail hemangioma there are thin walled vascular channels dissecting collagen fibers. Both lesions are currently thought to have these features due to trauma to a pre-existing vascular lesion [18, 21] and the papillary hyperplasia may persist after the evidence of trauma or thrombotic event has disappeared [21]. We believe that in our case of EAH the unusual vascular features may have been induced by trauma. This is supported by the patient's history and clinical findings (lower abdomen, changed lesions, evidence of belt pressure over biopsed lesions). In this particular case histology clearly separated EAH and other vascular malformations, such as those associated with blue rubber bleb nevus syndrome, and proved itself as a powerful diagnostic tool in this puzzling clinical cases.
Acknowledgment: We thank Mr Ricardo Moreira, Undergraduate Student of Department of Biomedical Science, University of Lincoln, UK for his help.
References
1. Nair L V, Kurien A M. Eccrine angiomatous hamartoma. Int J Dermatol. 1994; 33:650-1. [PubMed]2. Pinheiro AM, Ribeiro AM, Rodriques, HA. Eccrine angiomatous hamartoma: case report and clinical, pathologic and ultrasonographic studies. An Bras Dermatol. 2005; 80:377-80.
3. Martinelli P T, Tschen J A. Eccrine Angimatous Hamartoma: A Case Report and Review of the Literature. Cutis.2003; 71:449-455. [PubMed]
4. Smith VC, Montesinos E, Revert A, et al. Eccrine angiomatous hamartoma: report of three patients. Pediatr Dermatol.1996; 13:139-142. [PubMed]
5. SulicaRL, KaoGF, SulicaVI, et al. Eccrine angiomatous hamartoma (nevus): Immunohistochemical findings and review of the literature. Cutan Pathol. 1994; 21:71-75. [PubMed]
6. Lee HW, Han SS, Kang J, et al. Multiple mucinous and lipomatous variant of eccrine angiomatous hamartoma associated with spindle cell hemangioma: a novel collision tumor. J Cutan Pathol.2006:33:323-6. [PubMed]
7. Kaliyadan F, Sundeep V, Hiran KR, Fouzia Z. Late onset eccrine angiomatous hamartoma treated with intralesional sclerosant. Indian Journal of Dermatology. 2007; 52: 99-101.
8. Zeller DJ, Goldman RL. Eccrine-pilar angiomatous hamartoma: report of a unique case. Dermatologica. 1971: 143: 100-104. [PubMed]
9. Pelle MT, Pride HB, Tyler WB. Eccrine angiomatous hamartoma. J Am Acad Dermat. 2002: 47: 429-435. [PubMed]
10. Chicky Dadlani, Seth Orlov. Eccrine angiomatous hamartoma Dermatology Online Journal 2006 Sep 8; 12 (5):9 [PubMed]
11. Jeong E, Park HJ, Oh ST, et al. Late-onset eccrine angiomatous hamartoma on the forehead. Int J Dermatol (United States), May 2006, 45(5) :598-9 [PubMed]
12. Garcia-Arpa M, Rodriguez-Vazquez M, Cortina-de la Calle P, et al. Multiple and familial eccrine angiomatous hamartoma. Acta Derm Venereol (Norway), 2005, 85(4):355-7 [PubMed]
13. Sugimoto Y, Araki E, Inoguchi N, et al. Adult-onset multiple eccrine angiomatous hamartoma in enlarging hairy plaques.Acta Derm Venereol (Norway), 2007, 87(2):180-1 [PubMed]
14. Aloi F, Tomasini C, Pippione M. Eccrine angiomatous hamartoma: a multiple variant. Dermatology1992; 184(3):219-22 [PubMed]
15. Tanaka M., Shimizu S., Miyakawa S. Hypertrophic eccrine glands in eccrine angiomatous hamartoma produce gross cystic disease fluid protein 15. Dermatology 2000; 200:336-7 [PubMed]
16. Damiani S., Riccioni L. Palmar cutaneous hamartoma Am J Dermatopath 1998; 20:65-8 [PubMed]
17. Santa Cruz DJ, Aronberg J. Targetoid hemosiderotic haemangioma Am Acad Dermatol. 1988 Sep; 19(3):550-8.[PubMed]
18. Mentzel T, Partanen TA, Kutzner H. Hobnail hemangioma ("targetoid hemosiderotic hemangioma"): clinicopathologic and immunohistochemical analysis of 62 cases.J Cutan Pathol. 1999 Jul; 26(6):279-86.[PubMed]
19. Requena L, Sangueza OP. Cutaneous vascular proliferation. Part II. Hyperplasias and benign neoplasms.J Am Acad Dermatol. 1997 Dec; 37(6):887-919; [PubMed]
20. Franke FE, Steger K, Marks A, Kutzner H, Mentzel T. Hobnail hemangiomas (targetoid hemosiderotic hemangiomas) are true lymphangiomas. J Cutan Pathol. 2004 May; 31(5):362-7.[PubMed]
21. Clearkin KP, Enzinger FM. Intravascular papillary endothelial hyperplasia. Arch Pathol Lab Med. 1976 Aug; 100(8):441-4. [PubMed]
© 2009 Dermatology Online Journal