Localized cutaneous reaction to intramuscular vitamin K in a patient with acute fatty liver of pregnancy
Published Web Location
https://doi.org/10.5070/D32wp0r3bwMain Content
Letter: Localized cutaneous reaction to intramuscular vitamin K in a patient with acute fatty liver of pregnancy
Tatiana Sousa MD1, Lindsey Hunter BS2, Matthew Petitt DO3, Michael George Wilkerson MD3
Dermatology Online Journal 16 (12): 16
1. Department of Dermatology, Case Western Reserve-Metro Health Center, Cleveland, Ohio2. School of Medicine, University of Texas Medical Branch, Galveston, Texas
3. Department of Dermatology, University of Texas Medical Branch, Galveston, Texas. mgwilker@utmb.edu
Abstract
Vitamin K1 is frequently used in the treatment and prevention of hypoprothrombinemia and hemorrhagic disease of the newborn. It also serves as an antidote to anticoagulants. Erythematous, indurated, pruritic plaques uncommonly occur in adults after intramuscular injection with vitamin K1. We present a case of a localized cutaneous reaction to intramuscular vitamin K1 in a peripartum patient with acute fatty liver of pregnancy. The history and clinical presentation of our case is presented with a discussion of the pathogenesis pathogenesis of vitamin K1 and available treatment for this condition.
Introduction
Vitamin K is an essential cofactor for several clotting factors and anticoagulant proteins. Vitamin K1 (phylloquinone or phytonadione) is found in plants and Vitamin K2 (a menaquinone) is synthesized by Gram positive bacteria found in normal intestinal flora [1]. A deficiency of vitamin K results in symptoms of abnormal bleeding, including epistaxis, hematuria, ecchymoses, and bleeding from the gastrointestinal tract. Vitamin K, commonly in the form of phytonadione, is frequently used in the treatment and prevention of both hypoprothrombinemia and hemorrhagic disease of the newborn. In addition, it serves as an antidote to anticoagulants.
Although phytonadione is rather nontoxic, severe adverse reactions can infrequently occur [2]. Phytonadione is available in oral, intramuscular, or intravenous routes. With repeated injections, cutaneous reactions to vitamin K may arise and have been described in the literature. We report a case of a localized cutaneous reaction to intramuscular vitamin K in a peripartum patient with acute fatty liver of pregnancy complicated by kidney and liver dysfunction.
Case report
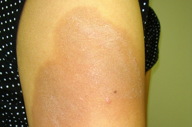 | 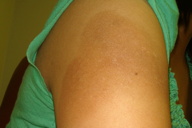 |
| Figure 1 | Figure 2 |
|---|
A 24-year-old G2P1A0 woman delivered a healthy infant at 38 weeks gestation. Four days prior, she experienced fevers, malaise, nausea and myalgias. Peripartum leukocytosis, hypoprothrombinemia, and abnormal hepatic and renal function were noted. Significant labs included: WBC 20.2 K/mm³, PT 20.2 sec, INR 3.03, AST 999, ALT 961, Alk Phos 607, total bilirubin 4.4, BUN 21, and Cr 2.7, with an estimated glomerular filtration rate of 23 mL/min/1.73 m². Multiple intramuscular (IM) injections of aqueous vitamin K1 (phytonadione) 10 mg/L for hypoprothrombinemia were given. Both hepatic and renal function improved postpartum. Eleven days later, the patient developed multiple well-demarcated, hyperpigmented, erythematous, eczematous plaques at injection sites (Figure 1). Fluticasone 0.05 percent cream and hydroxyzine were prescribed for pruritus. At a 4-week follow-up, her condition had improved, leaving residual post-inflammatory hyperpigmentation (Figure 2).
Discussion
Though infrequent, cutaneous reactions to vitamin K1 do occur. The pathogenesis is believed to be immune-mediated via a delayed (type IV) hypersensitivity reaction. Cutaneous reactions have been reproduced by patch testing [1]. Clinical presentations and morphology vary, with reactions consisting of erythematous, tender, indurated plaques, erythematous plaques or localized eczematous eruptions at the injection sites [2]. Localized urticaria and contact dermatitis can also occur [3]. Cutaneous reactions develop after 1-2 weeks and last 2-4 weeks. Rarely, patients experience a prolonged course with development of sclerotic plaques 2-4 months after treatment, lasting for years [4].
Histopathology demonstrates perivascular infiltrates with increased eosinophils and papillary dermal edema. Parakeratosis and spongiotic vesicles in the epidermis have been seen [2, 3]. Extension to the reticular dermis with a predominant mononuclear perivascular infiltrate can occur, although a neutrophilic infiltrate may rarely be seen [2].
Hypersensitivity reactions to vitamin K1 treatment were first described in patients with liver disease secondary to cirrhosis and/or hepatitis [5]. Recent information suggests this phenomenon is not restricted to those with liver dysfunction and is dose-independent [5]. Vitamin K1 hypersensitivity has also been reported in patients with autoimmune disorders, glomerulonephritis, amyloidosis, and hematologic disorders. Coexisting diagnoses of hepatic and autoimmune diseases might predispose patients to developing this delayed-type hypersensitivity reaction.
Finkelstein et al. reported a case involving a patient with systemic lupus erythematosus, admitted with preeclampsia and disseminated intravascular coagulation at 38 weeks gestation [6]. A similar case report discussed a patient admitted for labor; she received three IM injections of vitamin K1 and later developed a hot, red, pruritic plaque at the injection sites. This patient did not have hepatic disease or other coexisting pregnancy-related conditions [7]. To the best of our knowledge, our patient with vitamin K1 cutaneous hypersensitivity and acute fatty liver of pregnancy (AFLP) complicated by renal failure is the only such reported case. AFLP classically arises in the third trimester, with nonspecific symptoms, and characteristically elevated aminotransferase and bilirubin levels with a prolonged prothrombin time. Acute renal failure is common in these patients. Definitive treatment is delivery without delay.
Treatments for vitamin K1 hypersensitivity are not very effective and consist of topical corticosteroids, intralesional corticosteroids, oral antihistamines, topical tacrolimus, and pulsed dye laser [8, 9].
References
1. Wong DA, Freeman S. Cutaneous allergic reaction to intramuscular vitamin K1. Australas J Dermatol. 1999;40:174-152. [PubMed]2. Barnes HM, Sarkany I. Adverse skin reaction from vitamin K1. Br J Dermatol. 1976;95(6);653-656. [PubMed]
3. Bruynzeel I et al. Cutaneous hypersensitivity reactions to vitamin K: 2 case reports and a review of the literature. Contact Dermatitis. 1995;32(2):78-82. [PubMed]
4. Tuppal R, Tremaine R. Cutaneous eruption from vitamin K1 injection. J Am Acad Dermatol. 1992;27(1):105-106. [PubMed]
5. Moreau AC, Giordano LF, Bazex, J. [Cutaneous hypersensitivity at the site of injection of vitamin K1]. Ann Dermatol Venereol. 1996;123:177-179. [PubMed]
6. Finkelstein H, Champion MC, Adam JE. Cutaneous hypersensitivity to vitamin K1 injection. J Am Acad Dermatol. 1987;16:540-545. [PubMed]
7. Sanders MN, Winkelmann RK. Cutaneous reactions to vitamin K. J Am Acad Dermatol. 1988;19:699-704. [PubMed]
8. Wilkins K, DeKoven J, Assaad D. Cutaneous reactions associated with vitamin K1. J Cutan Med Surg. 2000;4(3):164-168. [PubMed]
9. Jung J et al. Recalcitrant Adverse Reaction to Vitamin K: Response to Pulsed Dye Laser. Dermatol Surg. 2004;30:931-933. [PubMed]
© 2010 Dermatology Online Journal

