Cidofovir
Published Web Location
https://doi.org/10.5070/D32vz9c4hzMain Content
A Review of Topical and Intralesional Cidofovir
Edward J. Zabawski, Jr.1, 2
Dermatology Online Journal 6(1): 3
1 Department of Dermatology Diagnostic Clinic of Longview, TX2Consultant in Dermatology Overton-Brooks VA Medical Center. Shreveport, LA
Abstract
Cidofovir is a potent nucleoside analog antiviral drug approved for the treatment of cytomegalovirus (CMV) retinitis in patients with the Acquired Immune Deficiency Syndrome (AIDS). It is currently available only for intravenous infusion. Several small studies and case reports describe the successful use of cidofovir applied either topically or by intralesional injection in several virally induced cutaneous diseases. Available information demonstrates that cidofovir is a potent antiviral agent with activity against several DNA viruses that cause cutaneous disease when applied topically or administered by intralesional injection. No significant systemic side effects have been noted, although application site reactions are common and can occasionally be severe. The effective use of topical and intralesional cidofovir for the treatment of diseases of the skin caused by DNA viruses has been demonstrated in animals and a limited number of patients including those infected with human immunodeficiency virus (HIV). This article reviews the pharmacology of cidofovir and the utility of topical and intralesional cidofovir for the treatment of viral infections caused by human papillomavirus, herpesviruses (including acyclovir resistant strains), Kaposi's sarcoma-associated herpesvirus, molluscum contagiosum and monkeypox.
Introduction
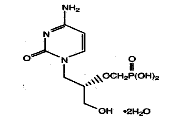 |
| Figure 1 |
|---|
| Figure 1. Cidofovir: [(S)-1-(3-hydroxy-2-phosphonylmethoxypropyl) cytosine; HPMPC, Vistide] |
Cidofovir is a nucleoside analog of deoxycytidine monophosphate (figure 1) that has antiviral activity against a broad range of DNA viruses.[
1] It is currently available for intravenous administration for the treatment of cytomegalovirus retinitis in AIDS patients. It is a water-soluble polar molecule that should be amenable to absorption across the gastrointestinal tract following oral administration although no oral formulations are currently available and bioavailability studies have been performed to a limited extent only in animals. It has been compounded in bases for topical use although topical formulations are prohibitively expensive (approximately $65 US per gram of extemporaneously compounded 3% cidofovir cream). This article briefly reviews the pharmacology, mechanism of action and uses for topical and intralesional cidofovir that have been described to date.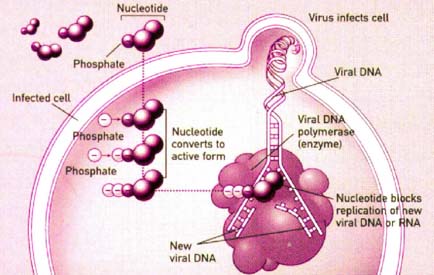 |
| Figure 2 |
|---|
| Figure 2. The mechanism of action of cidofovir in cells infected with cytomegalovirus. |
Pharmacology
Upon incorporation into the host cell, cidofovir (CDV) undergoes 2 stages of phosphorylation, via monophosphate kinase and pyruvate kinase, respectively, to form an active metabolite, cidofovir diphosphate (CDVpp) (figure 2). CDVpp bears structural similarity to nucleotides and acts as a competitive inhibitor and an alternative substrate for CMV DNA polymerase.[
2] It becomes incorporated into the growing CMV DNA strand and blocks further viral DNA synthesis leading to non-productive infection.[2] Incorporation of two consecutive molecules of CDVpp results in complete termination of DNA synthesis.[2] CDV inhibits viral DNA polymerase much more avidly than human DNA polymerase.[3] In addition, human CMV DNA polymerase is unable to excise incorporated CDVpp from viral DNA, which, in combination with reduction in viral DNA synthesis, may explain the prolonged activity of cidofovir [2] beyond the 17 to 65 hour half-life of the active metabolite.[3] Obviously, as it is incorporated into the growing DNA strand, CDV demonstrates no activity against RNA viruses.The mechanism of action in cells infected with HPV (human papillomavirus) apparently differs from that of CMV, as HPV utilizes host cell DNA polymerase instead of a virally encoded polymerase.[
4] While keratinocytes not infected with HPV show little or no decrease in growth when treated with cidofovir, infected cells show a marked decrease in cell growth.[5] These infected cells have been shown to be trapped in S phase, an indication of a halt of DNA synthesis.[4] CDV has been further demonstrated to induce DNA fragmentation and CPP32 (Caspase-3) protease activity, a key early event in the induction of apoptosis, in HPV positive cells.[4,6] This pronounced growth inhibitory effect of CDV on HPV infected cells includes malignant cell lines, as CDV has demonstrated both in vitro and in vivo activity against cervical carcinoma cells infected with HPV.[5,7,8] Activity against other virally induced tumors and hemangiomas has been demonstrated.[8]Unlike acyclovir and gancyclovir, CDV is not dependent upon virally encoded thymidine kinase for activation. Mendel et al determined that the antiviral activity of CDV against herpes simplex virus with mutant thymidine kinase is not only maintained but appears to be enhanced in tissue culture.[
9] Morphin et al showed that strains of herpesvirus that were resistant to acyclovir, gancyclovir and/or foscarnet remained sensitive to CDV.[10] Studies describing resistance of DNA viruses to cidofovir are few. Persistence of CMV DNA in the blood of AIDS patients is not thought to be due to CMV strains cross resistant to both CDV and gancyclovir or new cidofovir resistance.[11] In CMV strains with mutations conferring resistance to both CDV and gancyclovir, susceptibility to foscarnet was seen.[12] A study in an ocular animal model demonstrated that the antiviral activity of topical cidofovir is inactivated by concomitant application of topical corticosteroid.[13]The pharmacokinetic properties of CDV in humans have only been reported for the intravenous administration of this drug. CDV exhibits dose-independent pharmacokinetic parameters (i.e. volume of distribution, Cmax) when administered systemically.[
14] After a single dose, approximately 90% of CDV is recovered in the urine within 24 hours.[15] Because probenecid has been shown to reduce the renal clearance of CDV to that of creatinine clearance, elimination of CDV from the systemic circulation by active tubular secretion in addition to filtration has been suggested.[15] Some animal data regarding topical administration of CDV is available. Cundy et al investigated the bioavailability and metabolism of topical CDV in various vehicles on normal and abraded skin in rabbits.[16] The bioavailability of radiolabeled CDV on normal, intact skin was 0.2 to 2.1% and was enhanced in vehicles containing propylene glycol. The bioavailability of CDV on abraded skin in a vehicle containing propylene glycol was 41%. Topical application of cidofovir to intact skin in these animals led to negligible systemic exposure to the drug. Although no human studies have been performed to evaluate the extent that topical or intralesional cidofovir reaches the systemic circulation, rabbit skin is a reasonable model for comparison to humans. Consequently, application of CDV to skin with a damaged or absent epidermis should be avoided. The systemic side effects after intravenous administration of CDV are well established.[14] Nephrotoxicity, neutropenia and metabolic acidosis are not uncommon when CDV is administered intravenously.[14]In another study, Cundy et al observed the pharmacokinetic properties of CDV in African green monkeys by intravenous, oral and subcutaneous routes of administration.[
17] Of note, the subcutaneous bioavailability was 98 +/- 15.8%. In humans, systemic adverse reactions after topical or intralesional administration of CDV have only been reported in a single patient.[18] Snoeck et al reported that 1 of 11 patients treated for severe, recurrent laryngeal papillomatosis with intralesional cidofovir as a 2.5mg/ml solution withdrew from the protocol due to "precardial" complaints, presumably chest pain, after treatment.[18] No cardiac abnormalities were documented, however.Cidofovir is embryotoxic in both rats and rabbits at doses that were also toxic to the mother.[
14] An increased incidence of fetal soft tissue and skeletal anomalies was observed in rabbits treated intravenously at 1.0mg/kg/day.[14] Two separate studies in rats showed the development of mammary adenocarcinomas after systemic administration of CDV [14,19]. The appearance of malignant neoplasms has not been described in humans or other animal species after administration of CDV.[19] No studies regarding the administration of CDV by any route to pregnant women is available. The use of CDV in pregnant women should be avoided unless the benefit clearly outweighs the risk to the fetus.[14]Antiviral Effects
Human Papillomavirus Infections
Laryngeal Papillomatosis
Van Cutsem et al described the successful treatment of a large HPV-induced squamous papilloma of the hypopharynx and esophagus with intralesional cidofovir [
20]. A 69-year old woman with laryngeal papillomata unresponsive to interferon alpha and laser therapy was treated with 7 intralesional injections of CDV over a 5-month period. An endoscopic examination 7 months later failed to reveal residual lesions. No adverse effects were reported.Petersen and colleagues report the case of a 34-year old immunocompetent male with a history of laryngeal papillomas who had over 80 surgeries in 30 years [
21]. A dubious diagnosis of invasive carcinoma was made at age 20. At age 34, rapid progression was seen and interferon was tried without effect. Intralesional cidofovir was instituted at 2.5mg/ml once weekly, to a volume of 10-15 cc each treatment. While local regression was evident, invasive intracranial disease was discovered and the patient died.Snoeck et al treated seven females and ten males with severe, recurrent laryngeal papillomatosis with intralesional cidofovir as a 2.5mg/ml solution [
18]. Injections were performed on an in-patient basis every other week until improvement was seen, then less often. Fourteen of the seventeen patients showed complete remission, with 4 patients clearing initially, relapsing, then clearing again to achieve remission. One patient has been 27 months without recurrence. Of the remaining 3 patients, one has improved and now has stable disease, one progressed on cidofovir therapy after initial improvement, and one was lost to follow-up. Duration of treatment varied from 1 month to 13 months. Side effects were minimal, and no evidence of local necrosis or fibrosis was seen.Condyloma Acuminata
Snoeck et al first reported the use of topical CDV for relapsing anogenital condylomata in 3 patients with AIDS.[
22] A 44 year old homosexual man with penile lesions previously treated with curettage and podophilox was treated with 1% topical CDV once a day for 5 days. The lesions resolved and he remained free of lesions one year later. A 20 year old homosexual man with recurrent perigenital condylomata was also treated with 1% topical CDV. After 11 days, verrucous lesions were replaced with erosions that healed in 7 days. Six months later, no recurrence was evident. A 34 year old woman with recurrent condyloma acuminata of the vulva and perigenital skin was clear after treatment with 1% topical cidofovir gel applied once a day for 5 weeks. During 6 months of follow-up, she did not suffer a relapse.Douglas et al evaluated the use of topical cidofovir gel for the treatment of condyloma acuminata in patients with AIDS in phase I/II clinical trial.[
23] HIV positive patients with biopsy proven condyloma acuminata received open label cidofovir topical gel at 0.3%, 1% or 3% strengths over 5-or 10- day periods. Complete or partial responses were observed in 65% of patients (n= 46). All patients treated with 3% topical cidofovir for 10 days had some response. Although dose related mild to moderate application site reactions were seen in 39% of patients, no systemic toxicities were noted.Snoeck et al studied the use of cidofovir gel 1% in 19 immunocompetent patients of both sexes with biopsy proven condyloma acuminata.[
24] The lesions were untreated, refractory or recurrent. Sixteen of nineteen patients had at least a 50% reduction in wart area at 12 weeks, with nine patients demonstrating 100% clearance. Of complete responders, only one had a same site recurrence (median follow up: 180 days). While 65% experience application site side effects, no systemic toxicities were reported.Verruca Vulgaris
We reported 2 cases of verrucae vulgares refractory to conventional therapy that responded to treatment with topical CDV.[
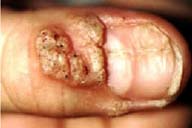
25] A seven-year old girl with several hundred verrucae of both legs had been treated with several different conventional therapies without success. Courses of topical 3% CDV cream twice a day for 10 days resulted in local inflammation and post-inflammatory hyperpigmentation followed by complete resolution of lesions. She has remained completely free of lesions for over 40 weeks. A 13-year old girl presented with verrucae of the distal fingers of both hands. Several conventional therapies including laser destruction had failed to yield lasting improvement. After treatment with cidofovir 3 % cream base once a day for 10 weeks, all lesions had resolved. Only minor local irritation was noted with no evidence of systemic toxicity. Twelve months following treatment, the patient has remained free of lesions.I have treated other pediatric patients with topical cidofovir in whom more conventional therapies were unsuccessful, contraindicated or declined (unpublished data). Even large lesions respond with significant clearing. In a case involving an 8-year-old boy, a large verruca affecting the dorsal surface of the thumb was present, as well as smaller lesions affecting the lateral nail folds (figure 3). The tumor was large enough to encroach on the underlying nail matrix, causing nail dystrophy.
Kaposi Sarcoma
In an in vitro study Kedes and Ganem evaluated the activity of various antiviral agents against Kaposi's sarcoma-associated herpesvirus (KSHV), now referred to more commonly as herpesvirus type 8. Cidofovir was found to be the most potent inhibitor of KSHV of the 4 agents tested (acyclovir, cidofovir, foscarnet, gancyclovir).[
26] Another in vitro study by Medveczky et al showed linear KSHV DNA synthesis and virus secretion to be very sensitive to cidofovir and moderately sensitive to gancyclovir or phosphonoformic acid, although episomal KSHV DNA synthesis was not inhibited by any agent.[24] Others have demonstrated the unique potency of cidofovir against KSHV.[28]Hammoud el al described the disappearance of a KaposiÌs sarcoma (KS) lesion in a 32-year-old AIDS patients treated with intravenous CDV.[
29] No change was noted on antiviral therapy with zidovudine, lamivudine and nelfinavir after 1 month, and only CDV weekly was added when the KS lesion decreased in size over 2 months. Simonart et al treated a patient with lesions of classic KS (HIV negative) with intralesional cidofovir with no effect.[30] Weekly injections of cidofovir 2.5 mg/cc were compared to saline and no difference could be detected. Concentrations of cidofovir from intralesional administration exceed those achieved with intravenous administration. Simonart, Snoeck and colleagues believe that HHV-8 represents a latent infection, and suggest that CMV triggers the release of cytokines and growth factors that result in KS.[31]Poxvirus Infections
Molluscum Contagiosum
Meadows et al reported 3 HIV seropositive patients with extensive molluscum contagiosum, 2 of whom received intravenous CDV and one of whom received topical CDV.[
32] One of the patients was a 26-year old HIV seropositive man who presented with extensive molluscum contagiosum lesions of the face, torso, arms and perianal area. Numerous attempts to chemically and surgically eradicate the lesions had failed. Intravenous CDV for treatment of resistant CMV retinitis was administered. Over 2 months of therapy, all of the molluscum contagiosum lesions resolved. A 31-year-old man with molluscum contagiosum also received intravenous cidofovir for treatment of CMV retinitis. Marked reduction in molluscum lesions was apparent by 2 weeks, and complete resolution of all lesions was noted at 4 weeks. Both patients remained clear of molluscum lesions while receiving maintenance intravenous cidofovir at the time of the report. A 37-year old man with extensive molluscum contagiosum of the face was treated with topical tretinoin and podophyllin solution with little success. Cidofovir 3% cream was initiated once a day, Monday through Friday, for 2 weeks. Moderate inflammation appeared during the second week of therapy. One month following therapy, neither molluscum contagiosum lesions nor residual inflammation were noted.Davies et al reported the successful treatment of a child with severe molluscum contagiosum infection and Wiskot-Aldrich syndrome.[
33] At age twelve, the patient had molluscum contagiosum lesions covering more than 75% of his body. Cidofovir 1% resulted in clearing of treated areas within 2-3 weeks. Acute inflammation was evident, but no systemic side effects were reported.Zabawski and Cockerell reported the successful treatment of two immunocompetent children with molluscum contagiosum with topical cidofovir.[
34] An 8-year-old girl and a 7-year-old boy both received cidofovir 3% cream twice daily. Though both had some objective site-specific irritation, no systemic adverse reactions were described. The lesions cleared in 6 weeks in the girl and 4 weeks in the boy, and no recurrence was noted at 6 months in each.Monkeypox
Although no human trials have been performed, cidofovir was found to decrease morbidity and mortality associated with monkeypox (orthopoxvirus) infections in 2 animal models.[
35] Single dose treatment protected mice as late as 5 days post-infection (mean time to death in controls 8-9 days). Primates treated with a single dose on the day of infection were completely protected from clinical and laboratory signs of disease.Herpesvirus Infections
Herpes Simplex Infection
De Clercq and Holy demonstrated that topical cidofovir was effective against human herpesvirus types 1 and 2 and thymidine kinase deficient herpesvirus type 1 in mice. They demonstrated that its efficacy was superior to acyclovir [
36].Snoeck et al reported the use of topical cidofovir in 2 patients with resistant herpesvirus infections.[
37] The first patient had AIDS and a chronic perineal HSV-2 infection that did not respond to acyclovir. Because the patient was intolerant to foscarnet, topical application of CDV 3 % gel daily was instituted. After 3 days of treatment, there was complete healing. After 3 weeks, the lesions recurred but resolved again with 3 days of daily application of CDV 3 % gel. At 7 weeks, there was another relapse but the virus was demonstrated to be sensitive to acyclovir and treatment with this agent resulted in resolution. The second patient was a bone marrow transplant recipient who developed severe oral HSV 1 infection resistant to both acyclovir and foscarnet. Two consecutive courses of topical CDV resulted in the emergence of an acyclovir susceptible strain that was treated successfully.Lateef et al described a 4-year-old child with AIDS with a large facial ulcer secondary to herpesvirus type 1 treated with topical cidofovir 1%.[
38] Viral cultures showed resistance to acyclovir, and treatment with intravenous foscarnet failed, as did topical trifluorothymidine. There was marked improvement with topical cidofovir 1 % over 2 weeks. The child died shortly after from complications related to AIDS.Lalezari et al reported a randomized, double blind, placebo controlled phase I/II clinical study of cidofovir gel in 30 patients with AIDS, all of whom had acyclovir-resistant herpes simplex infections.[
39] Eleven patients received 0.3% gel, nine patients received 1.0% gel and 10 were treated with placebo once a day for 5 days. Fifty percent of cidofovir patients had at least 50% improvement in infection in contrast with no improvement in the placebo patients. Thirty percent of the cidofovir treated patients had complete healing, compared with none of the placebo treated patients. The median time for negative viral cultures to be obtained from lesions in the cidofovir treated group was 2 days. Eighty seven per cent of CDV treated patients and no placebo treated patients developed negative viral cultures. Application site reactions occurred in 25 % of cidofovir-treated patients and in 20% of placebo-treated patients. Of the 6 patients treated with CDV who had complete healing, the response was sustained in 3.Finally, Sacks et al described the successful use of cidofovir topical gel in otherwise healthy patients with recurrent genital herpes infection.[
40] Ninety-six patients were randomized in a double blind, placebo controlled trial. All patients were confirmed by viral culture or serology as having recurrent genital herpes simplex. Treatment consisted of a single application of cidofovir gel 1%, 3%, 5% or placebo within 12 hours of an outbreak. All patients treated with cidofovir showed a decrease both in median time for cultures to become negative and in the number of days to complete healing. Sacks et al concluded that topical cidofovir gel was well tolerated and possessed significant antiviral activity.[40]An application to approve the use of topical cidofovir for acyclovir-resistant herpesvirus infections was denied by the United States Food and Drug Administration, citing the need for phase three data.[
19] According to the manufacturer, no further controlled studies to evaluate the use of topical cidofovir in herpes simplex infections are planned or in progress.[19]Conclusions
Cidofovir is an antiviral drug that has a broad spectrum of activity against a number of different DNA viruses. Continued investigations into its potential to treat malignant neoplasms and life threatening viral infections such as monekypox in addition to merely symptomatic viral maladies is to be encouraged. Its routine use in dermatology is hampered by its high cost, lack of continued interest by the manufacturer, and nagging concerns regarding carcinogenicity. The appearance of tumors in humans or animals other than rats has not been described, so that the true role of CDV as a carcinogen remains unclear. In contrast, its activity as a potent antiviral drug and, more importantly, as an antitumor drug is well established.[
30,31,34]References
1. De Clercq E. Therapeutic potential of Cidofovir (HPMPC, Vistide) for the treatment of DNA virus (i.e. herpes-, papova-, pox- and adenovirus) infections. Verh K Acad Geneeskd Belg 1996;58(1):19-47; discussion 47-9. PubMed2. Xiong X, Smith JL, Chen MS. Effect of incorporation of cidofovir into DNA by human cytomegalovirus DNA polymerase on DNA elongation. Antimicrob Agents Chemother 1997;41(3):594-9. PubMed
3. Ho HT, Woods KL, Bronson JJ, De Boeck H, Martin JC, Hitchcock MJ. Intracellular metabolism of the antiherpes agent (S)-1-[3-hydroxy-2-(phosphonylmethoxy)propyl]cytosine. Mol Pharmacol 1992;41(1):197-202. PubMed
4. Johnson JA, Gangemi JD. Selective inhibition of human papillomavirus-induced cell proliferation by (S)-1-[3-hydroxy-2-(phosphonylmethoxy)propyl]cytosine. Antimicrob Agents Chemother 1999;43(5):1198-205. PubMed
5. Andrei G, Snoeck R, Piette J, Delvenne P, De Clercq E. Antiproliferative effects of acyclic nucleoside phosphonates on human papillomavirus (HPV)-harboring cell lines compared with HPV-negative cell lines. Oncol Res 1998;10(10):523-31. PubMed
6. Andrei G, Snoeck R, Piette J, Delvenne P, De Clercq E. Inhibiting effects of cidofovir (HPMPC) on the growth of the human cervical carcinoma (SiHa) xenografts in athymic nude mice. Oncol Res 1998;10(10):533-9. PubMed
7. Andrei G, Snoeck R, Piette J, Delvenne P, De Clercq E. Inhibiting effects of cidofovir (HPMPC) on the growth of the human cervical carcinoma (SiHa) xenografts in athymic nude mice. Oncol Res 1998;10(10):533-9. PubMed
8. De Clercq E, Andrei G, Balzarini J, Hatse S, Liekens S, Naesens L, Neyts J, Snoeck R. Antitumor potential of acyclic nucleoside phosphonates. Nucleosides Nucleotides 1999;18(4-5):759-71. PubMed
9. Mendel DB, Barkhimer DB, Chen MS. Biochemical basis for increased susceptibility to Cidofovir of herpes simplex viruses with altered or deficient thymidine kinase activity. Antimicrob Agents Chemother 1995;39(9):2120-2. PubMed
10. Morphin F, Snoeck R, Andrei G, De Clercq E. Phenotypic resistance of herpes simplex virus 1 strains selected in vitro with antiviral compounds and combinations thereof. Antivir Chem Chemother 1996;7:270-275
11. Bowen EF, Cherrington JM, Lamy PD, Griffiths PD, Johnson MA, Emery VC. Quantitative changes in cytomegalovirus DNAemia and genetic analysis of the UL97 and UL54 genes in AIDS patients receiving cidofovir following ganciclovir therapy. J Med Virol 1999;58(4):402-7. PubMed
12. Schmit I, Boivin G. Characterization of the DNA polymerase and thymidine kinase genesof herpes simplex virus isolates from AIDS patients in whom acyclovirand foscarnet therapy sequentially failed. J Infect Dis 1999;180(2):487-90. PubMed
13. Romanowski EG, Araullo-Cruz T, Gordon YJ. Topical corticosteroids reverse the antiviral effect of topical cidofovir in the Ad5-inoculated New Zealand rabbit ocular model. Invest Ophthalmol Vis Sci 1997;38(1):253-7. PubMed
14. Package insert. Vistide(cidofovir injection).Gilead Sciences. Inc., 1996.
15. Cundy KC, Petty BG, Flaherty J, Fisher PE, Polis MA, Wachsman M, Lietman PS, Lalezari JP, Hitchcock MJ, Jaffe HS. Clinical pharmacokinetics of cidofovir in human immunodeficiency virus-infected patients. Antimicrob Agents Chemother 1995;39(6):1247-52. PubMed
16. Cundy KC, Lynch G, Lee WA. Bioavailability and metabolism of cidofovir following topical administration to rabbits. Antiviral Res 1997;35(2):113-22. PubMed
17. Cundy KC, Li ZH, Hitchcock MJ, Lee WA. Pharmacokinetics of cidofovir in monkeys. Evidence for a prolonged elimination phase representing phosphorylated drug. Drug Metab Dispos 1996;24(7):738-44. PubMed
18. Snoeck R, Wellens W, Desloovere C, Van Ranst M, Naesens L, De Clercq E, Feenstra L. Treatment of severe laryngeal papillomatosis with intralesional injections of cidofovir [(S)-1-(3-hydroxy-2-phosphonylmethoxypropyl)cytosine]. J Med Virol 1998;54(3):219-25. PubMed
19. Written communication, J Buchanan, Pharm.D, Gilead Pharmaceuticals, Foster City, CA, April 1, 1997.
20. Van Cutsem E, Snoeck R, Van Ranst M, Fiten P, Opdenakker G, Geboes K, Janssens J, Rutgeerts P, Vantrappen G, de Clercq E, et al. Successful treatment of a squamous papilloma of the hypopharynx-esophagus by local injections of (S)-1-(3-hydroxy-2-phosphonylmethoxypropyl)cytosine. J Med Virol 1995;45(2):230-5. PubMed
21. Petersen BL, Buchwald C, Gerstoft J, Bretlau P, Lindeberg H. An aggressive and invasive growth of juvenile papillomas involving the total respiratory tract. J Laryngol Otol 1998;112(11):1101-4. PubMed
22. Snoeck R, Van Ranst M, Andrei G, De Clercq E, De Wit S, Poncin M, Clumeck N. Treatment of anogenital papillomavirus infections with an acyclic nucleoside phosphonate analogue [letter] N Engl J Med 1995;333(14):943-4. PubMed
23. Douglas J, Corey T, Tyring S, Kreisel J, Bowden B, Crosby D, Berger T, Conant M, McGuire B, Jaffe HS. A phase I/II study of cidofovir topical gel for refractory condyloma acuminatum in patients with HIV infection. Poster presented at the 4th conference on Retroviruses and Opportunistic Infections, Washington DC, January 22-26, 1997. Poster #334.
24. Snoeck R, Bossens M., Parent D., Delaere B., De Greef H., De Clercq, S.Safrin, B. McGuire, Jaffe H.S. . A Double-Blind, Placebo-Controlled Study of Cidofovir Gel for Human Papillomavirus (HPV)-Associated Genital Warts. Poster presented at the 11th International Conference on Antiviral Research, San Diego, California, April 5-10, 1998. Abstract #27.
25. Zabawski EJ Jr, Sands B, Goetz D, Naylor M, Cockerell CJ. Treatment of verruca vulgaris with topical cidofovir [letter] JAMA 1997;278(15):1236. PubMed
26. Kedes DH, Ganem D. Sensitivity of Kaposi's sarcoma-associated herpesvirus replication to antiviral drugs. Implications for potential therapy. J Clin Invest 1997;99(9):2082-6. PubMed
27. Medveczky MM, Horvath E, Lund T, Medveczky PG. In vitro antiviral drug sensitivity of the Kaposi's sarcoma-associated herpesvirus. AIDS 1997;11(11):1327-32. PubMed
28. Neyts J, De Clercq E. Antiviral drug susceptibility of human herpesvirus 8. Antimicrob Agents Chemother 1997;41(12):2754-6. PubMed
29. Hammoud Z, Parenti DM, Simon GL. Abatement of cutaneous Kaposi's sarcoma associated with cidofovir treatment [see comments] Clin Infect Dis 1998;26(5):1233. PubMed
30. Simonart T, Noel JC, De Dobbeleer G, Parent D, Van Vooren JP, De Clercq E, Snoeck R. Treatment of classical Kaposi's sarcoma with intralesional injections of cidofovir: report of a case. J Med Virol 1998;55(3):215-8. PubMed
31. Simonart T, Noel JC, De Clercq E, Snoeck R. Abatement of cutaneous Kaposi's sarcoma associated with cidofovir treatment [letter; comment] Clin Infect Dis 1998;27(6):1562. PubMed
32. Meadows KP, Tyring SK, Pavia AT, Rallis TM. Resolution of recalcitrant molluscum contagiosum virus lesions in human immunodeficiency virus-infected patients treated with cidofovir [see comments] Arch Dermatol 1997;133(8):987-90. PubMed
33. Davies EG, Thrasher A, Lacey K, Harper J. Topical cidofovir for severe molluscum contagiosum [letter] Lancet 1999;353(9169):2042. PubMed
34. Zabawski EJ Jr, Cockerell CJ. Topical cidofovir for molluscum contagiosum in children [letter] Pediatr Dermatol 1999;16(5):414-5. PubMed
35. Huggins JW, Smee D, Martinez M, Bray M. Cidofovir(HPMPC) Treatment of Monkeypox. Abstract presented at the 11th International Conference on Antviral Research, San Diego, California, April 5-10, 1998. Abstract #118
O36. De Clercq E, Holy A. Efficacy of (S)-1-(3-hydroxy-2-phosphonylmethoxypropyl)cytosine in various models of herpes simplex virus infection in mice. Antimicrob Agents Chemother 1991;35(4):701-6. PubMed
37. Snoeck R, Andrei G, Gerard M, Silverman A, Hedderman A, Balzarini J, Sadzot-Delvaux C, Tricot G, Clumeck N, De Clercq E. Successful treatment of progressive mucocutaneous infection due to acyclovir- and foscarnet-resistant herpes simplex virus with (S)-1-(3-hydroxy-2-phosphonylmethoxypropyl)cytosine (HPMPC). Clin Infect Dis 1994;18(4):570-8. PubMed
38. Lateef F, Don PC, Kaufmann M, White SM, Weinberg JM. Treatment of acyclovir-resistant, foscarnet-unresponsive HSV infection with topical cidofovir in a child with AIDS [letter; comment] Arch Dermatol 1998;134(9):1169-70. PubMed
39. Lalezari J, Schacker T, Feinberg J, Gathe J, Lee S, Cheung T, Kramer F, Kessler H, Corey L, Drew WL, Boggs J, McGuire B, Jaffe HS, Safrin S. A randomized, double-blind, placebo-controlled trial of cidofovir gel for the treatment of acyclovir-unresponsive mucocutaneous herpes simplex virus infection in patients with AIDS. J Infect Dis 1997;176(4):892-8. PubMed
40. Sacks SL, Shafran SD, Diaz-Mitoma F, Trottier S, Sibbald RG, Hughes A, Safrin S, Rudy J, McGuire B, Jaffe HS. A multicenter phase I/II dose escalation study of single-dose cidofovir gel for treatment of recurrent genital herpes. Antimicrob Agents Chemother 1998;42(11):2996-9. PubMed
© 2000 Dermatology OnLine Journal