Linear IgA bullous dermatosis: the more frequent bullous dermatosis of children
Published Web Location
https://doi.org/10.5070/D32ts975m4Main Content
Linear IgA bullous dermatosis: The more frequent bullous dermatosis of children
Monia Kharfi1, Aida Khaled1, Amel Karaa1, Ines Zaraa2, Becima Fazaa2, Mohamed Ridha Kamoun1
Dermatology Online Journal 16 (1): 2
1. Dermatology Department, Charles Nicolle Hospital, Tunis, Tunisia. aida.khaled@rns.tn2. Dermatology Department, La Rabta Hospital, Tunis, Tunisia
Abstract
Background: Linear IgA bullous dermatosis (LAD) of children is relatively frequent in Africa. We undertook this study to evaluate the frequency of this disease among autoimmune bullous diseases (AIBD) in Tunisian children. Methods: We present a 32-year retrospective study (January 1976 to December 2007). Children with chronic acquired bullous diseases seen at the Charles Nicolle Hospital of Tunis and for whom direct immunofluorescence (DIF) of the perilesional skin demonstrated linear IgA immunoglobulin deposits were included in the study population. Results: Thirty-one children with LAD were collected representing 65.9 percent of all AIBD of children collected in the same period, with a mean age of 5.5 years and a sex ratio M/F of 2.4. Most of the children had a generalized eruption (28/31) but more profuse on the face, pelvic region, buttocks, and limbs. Mucosal lesions were present in only 4 children (12.9%). The mean duration of the disease was 14 months. Direct immunofluorescence demonstrated predominantly linear IgA deposits along the dermal-epidermal junction in all patients. Faint IgG, IgM, and complement were also seen (20/31). Indirect immunofluorescence was negative in 67 percent of cases. Eight patients responded to Dapsone, but prednisone had to be added in 7 children and erythromycin in 4 others to control the disease. A long term remission period (34 months) was achieved in 61.9 percent of patients. Conclusion: This study confirms that LAD is the most common AIBD in children in Tunisia and it frequently occurs in preschool-aged males. Independently of the medication chosen for treatment, a long term remission is frequently observed.
Background
Linear IgA dermatosis (LAD) is a rare acquired autoimmune subepidermal vesiculo-bullous disease characterized by continuous linear IgA deposits in the basement membrane zone (BMZ), visualized on direct immunofluorescence microscopy. The clinical presentation is heterogeneous and can mimic other blistering diseases such as dermatitis herpetiformis and bullous pemphigoid. Linear IgA dermatosis can affect both adults and children; in the latter the disease is historically referred to as chronic bullous dermatosis of childhood (CBDC). The two forms, which differ slightly in their clinical presentations, have identical immunopathological and immunogenetic features. The frequency of the disease differs in children and adults and also from one country to another. Linear IgA dermatosis is the most frequent bullous dermatosis of children in France, Asia, and South Africa. There are very few epidemiologic studies of children with this distinct childhood disorder [1]. The aim of the present study is to highlight the epidemiological and clinical features and response to treatment in children with LAD attending our tertiary care Hospital in Tunis over a period of 32 years.
Methods
All children under 16 years of age and followed-up for blistering diseases at the Department of Dermatology of Charles Nicolle Hospital of Tunis over the past 32 years (January 1976 to December 2007) were studied. Our center is a tertiary care center serving a 500,000 population. This study was done after permission of our hospital internal review board.
Only children who showed evidence for predominant linear IgA immunoglobulin deposits in perilesional skin on direct immunofluorescence (DIF) were included in the study group and analyzed with the diagnosis of linear IgA dermatosis. In addition to demographic data, details regarding the age at onset, duration, and distribution of lesions, treatment, and outcome (remission under treatment and recurrence) were recorded.
Results
During the analyzed period, 47 children of all ages had been treated for a confirmed bullous dermatosis. Linear IgA dermatosis was by far the most common disease with 31 cases (65.9%). The other cases were distributed as follows: 8 cases of bullous pemphigoid, 5 cases of dermatitis herpetiformis, and only 3 cases of pemphigus.
Epidemiological, clinical, histological and immunopathological characteristics of the 31 patients are summarized in Table 1.
In children with LAD, the disease began at a mean age of 5.5 years old [range: 8 months - 16 years]. Both sexes were affected, but males outnumbered females in a ratio F/M of 2.4.
Most of the children gave a history of a sudden onset of the bullous eruption (14/31). The other patients noted a period of prodromal itching or transient mild pruritus before specific lesions erupted.
Only one of them had an intense pruritus with a burning sensation. Differentiation from an infectious bullous dermatosis (impetigo) was difficult in 8 children; in these physical examination was misleading and resulted in a delayed diagnosis.
 |  |
| Figure 1 | Figure 2 |
|---|---|
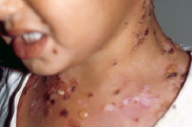
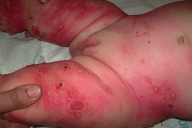
| Figure 1. Tense bullae with crusted erosions Figure 2. Vesicles and Bullae arising on normal appearing skin, or at the periphery of annular erythema | |
 |  |
| Figure 3 | Figure 4 |
|---|---|
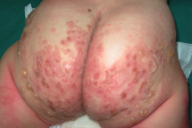
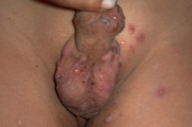
| Figure 3. Diffuse bullous eruption in an infant Figure 4. Genital involvement in a boy | |
In all children treated for LAD, we noted a significant polymorphism in cutaneous lesions with coexistence of several morphologies, at different stages of the disease. In all patients there were tense pruritic blisters of variable sizes with small clear-filled vesicles in some of them (12/31) (Figures 1, 2, 3, and 4). Bullae arose on normal appearing skin, or at the periphery of an annular erythema (in 15 patients) (Figure 2). Twelve of 31 patients had a cluster of jewels appearance, with lesions in a rosette pattern (Figure 2). Crusts, excoriations, and erosions were also present in some of the children (Figure 1). Seven children had lesions mimicking dermatitis herpetiformis with small-grouped blisters. Nine children (29%) had features of bullous pemphigoid with large and tense bullae. The rest of the children had a mixed pattern of both groups. Most of the children had a generalized eruption (28 children, 93%), with an asymmetrical distribution. Two patients had localized lesions of the legs. In 70 percent of cases, the lesions were more profuse on the perineum and particularly on the genital areas and buttocks, especially in boys (12 out of 13 cases, Figure 4). Perianal lesions were noticed in only one child. The face (chin and helix) was frequently involved (53% of the cases).
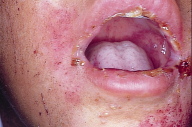
Mucous membrane involvement (oral and genital) was noticed in only 4 boys (12.9%) (Figure 5).
 |  |
| Figure 5 | Figure 6 |
|---|---|
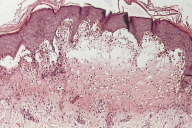
| Figure 5. Buccal erosions and facial involvement Figure 6. Subepidermal bulla with eosinophils and neutrophils and papillary abscesses | |
 |
| Figure 7 |
|---|
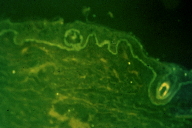
| Figure 7. Direct immunofluorescence with linear IgA deposits along the dermal epidermal junction |
Histological features showed in all patients a subepidermal bulla with eosinophils and neutrophils and papillary abscesses (Figure 6).
In all patients, DIF demonstrated predominant linear IgA deposits along the dermal epidermal junction (Figure 7). Twenty out of 31 children showed evidence of faint deposits of IgG, IgM and complement associated with IgA.
Indirect immunofluorescence (IIF) was negative in 67 percent of the patients (Table 1).
Malabsorption tests were conducted in 3 patients with no digestive signs; but one patient had partial jejunal atrophy.
HLA studies showed B8, DR3 antigen pattern in 2 of the 3 patients studied.
In our series, dapsone was used as a first line therapy in 70 percent of cases (19 children). The initial dose varied from 25 mg to 100 mg/day; the started dose was chosen on the basis of the patient's age and weight and the progression and severity of the disease. Table 2 summarizes data concerning treatment and outcome in 21 followed-up children.
Dapsone was used as a monotherapy in 8 cases and led to a partial or a complete healing of lesions in all of them, after a mean duration of 13 days [extremes: 3 and 23 days]. Treatment with dapsone was maintained at the same initial dose for a mean duration of 6.5 months [extremes: 3 and 21 months]. Five of these 8 patients had no flares during treatment and remained in remission even after stopping dapsone, with a mean remission period of 2.5 years [extremes: 4 months and 6 years]. One patient had continued to flare and then was lost to follow-up. The two other patients were lost to follow-up.
In 11 patients, because of an initial resistance to therapy, dapsone was combined with either oral corticosteroids (7 patients) (0.5 - 2 mg/Kg/day) or with erythromycin (4 patients).
The associated therapy (dapsone-oral steroids) produced healing in a mean period of 12 days [8-15 days]. Corticosteroids were then tapered to a maintenance dose of 5 to 15 mg/day, over a mean period of 20 months [extremes: 3 months and 6 years]. Localized relapses were observed during treatment in 5 of them. Six of these 7 patients (1 patient was lost to follow-up) had long standing remissions (off treatment) of a mean period of 3 years [extremes: 9 months and 11 years].
One of the 4 patients receiving erythromycin combined with dapsone had achieved remission with a 4 year-follow-up period.
Two patients had completely cleared with antibiotics: amoxicillin only (1 g/day) for one of them and oral sulfonamide antibiotic for the other (3 months and 2 years remission, respectively). Ten of the 31 diagnosed patients with LAD were lost to follow up. Independently of the drug used, a total remission was observed in 13 patients (61.9%) with a 34 month-follow-up period after stopping treatment [extremes 3 months-6 years]. Eight patients were lost to follow-up after the onset of treatment. Complications included methemoglobinemia and anemia in three children treated by dapsone, leading to a tapering of the doses, with a continued good response. In one patient, DIF performed in the remission period (4 years after stopping dapsone) was negative, testifying to a histological cure.
Discussion
Linear IgA dermatosis (LAD) is an autoimmune disease characterized immunopathologically by the linear deposition of IgA at the basement membrane zone. This antibody deposition leads to complement activation and to a cascade of immunoreactions resulting in the loss of adhesion at the dermal-epidermal junction; the major consequence is blister formation [2].
Linear IgA dermatosis is not a very common disease in adults. Its prevalence has been estimated in Europe as 0.5 per 1,000,000 adults [3]. The prevalence in children has rarely been reported but its incidence is reported to be high in South Africa, North Africa and Asia [1, 4, 5, 6]. Taking into account the previous reported series, our series describing 31 cases of LAD in 32 years is to our knowledge, among the highest number ever reported [5, 6]. It shows that LAD is the most common autoimmune bullous dermatosis (AIBD) in children in Tunisia with up to 66 percent.
Linear IgA dermatosis has a bimodal age of onset because it may have its onset in children and in adults. Disease in children begins usually in pre-school age, with a mean of 4-5 years [6, 7]. In our series the mean age at onset was in agreement with other reports. The disease affects both sexes, but some studies reported a slight female preponderance [8]. Our study and another recently reported Tunisian series, report an increased involvement of boys [5, 6, 9]. The latter study was undertaken at another Tunisian department serving the south and central population. There are no overlapping cases between their series and ours [9].
The disease has been associated most commonly with infections and drugs. Drug-induced LAD is more likely to occur in the older population with a significant evidence for association with vancomycin [10, 11]. Drug-induced LAD is exceptionally described in children [12]. In a series of 25 cases of childhood LAD reported by Wojnarowska et al., 38 percent of them had either preceding infection or ingestion of drugs. Drugs that have been associated with childhood LAD include antibiotics and non-steroidal inflammatory agents [13]. In our series, no causes have been implicated in the onset of the disease.
Clinically there are no major differences between the adult and the childhood forms of LAD. Only the distribution of lesions differs. In fact, lesions in children are typically localized in the lower abdomen and ano-genital areas with frequent involvement of the perineum. The other sites include face, especially the peri-oral area. Lesions may be symmetrically or asymmetrically distributed [8].
Mucosal involvement is more frequent in adults than in children particularly in Europe with oral, ocular, genital, and digestive manifestations [13, 14]. In African and Asian series, children presenting with LAD, rarely suffer from mucosal involvement [5, 6, 15]. This is confirmed in our study in which only 12.9 percent of children had evidence of oral or genital lesions and also by another recent Tunisian study [9]. But, even if no signs of mucosal affection are reported by the patient, an ocular examination is necessary because of the risk of subconjunctival fibrosis, symblepharon formation, and cicatricial entropion.
The association of LAD with a gluten-sensitive enteropathy is less frequently observed than with dermatitis herpetiformis (DH) and remains controversial because of the absence of clinical signs and the usual negativity of complementary examinations [2]. None of our patients had an abnormal digestive history but one had jejunal atrophy on endoscopy.
Linear IgA dermatosis in children has been reported in association with HLA B8 DR3 Cw7 more frequently than in adults. In addition, the disease is more likely, if these genes are present in a homozygote state [2]. Moreover, the TNF gene is thought to play a role in the duration of the disease and is associated with a worse prognosis [14]. In our series, two of the three children investigated for HLA haplotype, had B8, DR3 haplotype.
Immunofluorescence studies are very important for the diagnosis of LAD. DIF of perilesional and normal skin shows typically a predominant linear deposition of IgA along the basement membrane [8]. Complement deposition is frequent (C3) and IgG or IgM can also be present at the same time as IgA antibodies. In some cases, the DIF can be negative and the dermatosis can be mistaken for an infectious disease leading to a considerable delay in the treatment. Repeated laboratory studies must be considered in cases with obvious clinical presentation of LAD and negative DIF.
Circulating IgA BMZ antibodies have been reported in up to 80 percent of patients [5]. But IIF seems to be more likely to be positive in children than in adult patients with LAD (72% vs 20%) [16].
Mechanism of loss of self-tolerance to target antigens is unknown. Many studies have focused on the involved dermal-epidermal junction antigens and different protein targets have been identified, including the lamina lucida, sublamina densa or both locations simultaneously. The 97 kDa and the 120 kDa proteins extracted from human epidermis are the best characterized antigens binding IgA antibodies from sera of patients with LAD. They are localized in the lamina lucida. These proteins may represent a cleaved portion of the extracellular domain of bullous pemphigoid antigen (180 kDa), which can exist in vivo, or could also be alternative splicing products of the same antigen.
More recently, patients with LAD have been reported to show evidence of IgA antibodies reacting with both bullous pemphigoid antigens (180 kDa and 230 kDa). This may be explained by a specific immune response against pemphigoid antigens [2, 17].
A minority of patients can present with antibodies directed against different antigens located on both lamina lucida and lamina densa. The 280 kDa antigen, the collagen VII antigen (250 kDa) and many more, can be identified in those patients and can lead to the incorrect diagnosis of bullous pemphigoid (BP) or epidermolysis bullosa acquisita. More confusing, are the patients presenting with both IgA and IgG antibodies targeting the same antigens.
Independent of the targeted antigen, patients with predominantly linear IgA antibodies continue to be grouped into a single category of disease, that is LAD. On the other hand, and because of the molecular heterogeneity, patients can be classified as having BP with faint IgA deposits, epidermolysis bullosa acquisita with faint IgA deposits, and the IgA/IgG linear dermatosis [18, 19].
Our series, along with several other reported series (including the recent Tunisian one), confirm the efficiency of dapsone in controlling LAD. Excellent initial responses to treatment (no flares during the treatment) and long term remissions (2.5 years in 5 out of 8 patients of our series treated by dapsone) are common [5, 6, 9, 20].
However, a combination with systemic steroids is sometimes required to control the disease (seven patients in our series). In our patients, combination therapy with dapsone and oral prednisone led also to long term remission (mean=3 years).
Other useful medications including colchicine, niacinamide, antibiotics, and gluten free diet have been reported [2, 20]. Two of our patients had responded to antibiotic therapy only (sulfonamide and amoxicillin). Exclusive antibiotic therapy was also successful in five cases of the recently above-mentioned Tunisian series [9].
Local care of lesions must be conducted; ruptured bullae and infected lesions should be treated with sterile dressings and close observation. Attention to personal hygiene must be emphasized in children with perineal involvement.
Linear IgA dermatosis of childhood is usually a benign and self-limiting disorder; a longstanding remission can be achieved either with dapsone or a combination therapy [5, 6].
Sixty two percent of patients (13 over 21 followed patients) had a mean remission of 34 months after stopping the treatment.
The disease tends to wax and wane in severity over the years until it disappears spontaneously (this was the case in 9 of our patients, the remaining patients had a sustained remission even after stopping the treatment) [13]. The mean duration of the disease is usually 3 to 4 years, but long term involvement can also be observed. Cutaneous lesions heal without scarring if not infected or excoriated.
As demonstrated by one of our cases, IgA deposits on DIF may disappear completely in children after remission, but they generally remain positive in adults even after healing [2].
In conclusion, our study was in agreement with the occidental literature for some clinical features but it differs in the frequency of the disease, the male predominance, the rare mucosal involvement, and the shorter mean duration of activity with rapid response to treatment. These differences were also reported by other authors from Tunisian, African and Asian countries [4, 5, 9].
References
1. Denguezli M, Ben Nijma B, Nouira R, et al. IgA linear bullous dermatosis in children. A series of 12 Tunisian patients. Ann Dermatol Venereol 1994; 121:888-892. [PubMed]2. Guide S V, Marinkovich M P. Linear Ig A bullous dermatosis. Clin Dermatol. 2001;19: 719-27. [PubMed]
3. Zillikens D, Wever S, Roth A, Weidenthaler-Barth B, Hashimoto T, Brocker EB. Incidence of autoimmune subepidermal blistering dermatoses in a region of central Germany. Arch Dermatol. 1995; 131: 957-8. [PubMed]
4. Aboobaker J, Wojnarowska FT, Bhogal B, Black MM. Chronic bullous dermatitis of childhood-clinical and immunological features seen in African patients. Clin Exp Dermatol. 1991; 16:160-4. [PubMed]
5. Kanwar AJ, Sandhu K, Handa S. Chronic bullous dermatosis of childhood in north India. Pediatr Dermatol. 2004; 21: 610-2. [PubMed]
6. Nanda A, Dvorak R, Al-Sabah H, Alsaleh QA. Linear IgA bullous disease of childhood: an experience from Kuwait. Pediatr Dermatol. 2006; 23: 443-7. [PubMed]
7. Hruza LL, Mallory SB, Fitzgibbons J, Mallory GB Jr. Linear IgA bullous dermatosis in a neonate. Pediatr Dermatol. 1993; 10: 171-6. [PubMed]
8. Brenner S, Mashiah J. Autoimmune Blistering diseases in children: signposts in the process of evaluation. Clin dermatol. 2000; 18: 711-24. [PubMed]
9. Kenani N, Mebazaa A, Denguezli M, Ghariani N, Sriha B, Belajouza C, Nouira R. Childhood linear IgA bullous dermatosis in Tunisia. Pediatr Dermatol 2009; 26(1):28-33. [PubMed]
10. Kuechle MK, Stegemeir E, Maynard B, Gibson LE, Leiferman KM, Peters MS. Drug-induced linear Ig A bullous dermatosis: report of six cases and review of the literature. J Am Acad Dermatol 1994; 30: 187-92. . [PubMed]
11. Nousari HC, Kimyai-Asadi A, Caeiro JP, Anhalt GJ. Clinical, demographic, and immunohistologic features of vancomycin-induced linear IgA bullous disease of the skin. Report of 2 cases and review of the literature. Medicine (Baltimore). 1999; 78:1-8. [PubMed]
12. Ho JC, Ng PL, Tan SH, Giam YC. Childhood linear IgA bullous disease triggered by amoxicillin-clavulanic acid. Pediatr Dermatol. 2007; 24: 40-3. [PubMed]
13. Wojnarowska F, Marsden RA, Bhogal B, Black MM. Chronic bullous disease of childhood, childhood cicatricial pemphigoid, and linear IgA disease of adults. A comparative study demonstrating clinical and immunopathologic overlap. J Am Acad Dermatol. 1988; 19: 792-805. [PubMed]
14. Collier PM, Wojnarowska F, Welsh K, McGuire W, Black MM. Adult linear Ig A disease and chronic bullous disease of childhood: the association with human lymphocyte antigens cw7, B8, DR3 and tumor necrosis factor influences disease expression. Br J Dermatol. 1999; 141: 867-75. [PubMed]
15. Mahe A, Flageul B, Bobin P. Bullous IgA linear dermatosis of children in Mali. Ann Dermatol Venereol. 1996; 123: 544-8. [PubMed]
16. Bhogal B, Wojnarowska F, Marsden RA, Das A, Black MM, McKee PH. Linear Ig A bullous dermatosis of adults and children: an immunoelectron microscopic study. Br J Dermatol. 1987; 117 : 289-96. [PubMed]
17. Ghohestani RF, Nicolas JF, Kanitakis J, Claudy A. Linear Ig A bullous dermatosis with Ig A antibodies exclusively directed against the 180- or 230-kDa epidermal antigens. J Invest Dermatol. 1997; 108: 854-8. [PubMed]
18. Egan CA, Zone JJ. Linear Ig A bullous dermatosis. Int J Dermatol. 1999; 38: 818-27. [PubMed]
19. Sobjanek M, Sokolowska-Wojdylo M, Sztaba-Kania M, Baranska-Rybak W, Maciejewska A, Wlodarkiewicz A. Clinical and immunopathological heterogeneity of 22 cases of linear IgA bullous dermatosis. J Eur Acad Dermatol Venereol 2008; 22 (9):1131. [PubMed]
20. Alajlan A, Al-Khawajah M, Al-Sheikh O, Al-Saif F, Al-Rasheed S, Al-Hoqail I, Hamadah IR. Treatment of linear IgA bullous dermatosis of childhood with flucloxacillin. J Am Acad Dermatol. 2006; 54: 652-6. [PubMed]
© 2010 Dermatology Online Journal

