Calcipotriol and PUVA as treatment for vitiligo
Published Web Location
https://doi.org/10.5070/D32fk376frMain Content
Calcipotriol and PUVA as treatment for vitiligo
F Cherif1, MI Azaiz1, A Ben Hamida2, A Ben Osman Dhari1
Dermatology Online Journal 9 (5): 4
1. Department of Dermatology, La Rabta Hospital, Tunis, Tunisia.2. Department of Preventive Medicine, University of Tunis,
Tunisia. faika.cherif@voila.fr
Abstract
We performed a prospective study to evaluate efficacy of the combination of calcipotriol and psoralen plus ultraviolet A (PUVA) in the treatment of vitiligo. Twenty-three patients with essentially bilateral symmetrical lesions of vitiligo were included. Calcipotriol (0.005 %) ointment was applied twice daily over the right side of the body, and the other side was not treated. PUVA was performed three times per week. All patients received at least fortyfive sessions of PUVA. Patients were evaluated clinically and photographed all fifteen weeks. At the fifteenth session, 69 percent of the patients had minimal to moderate improvement on the calcipotriol side compared to 52 percent on the PUVA-only side (p = 0.015). At the forty-fifth session, 52 percent showed marked improvement on the calcipotriol side compared to 30 percent on the PUVA-only side (p = 0.13), with more intense repigmentation on calcipotriol-treated areas. Treatment was well tolerated, and no adverse effect was noted. This combination was an effective treatment for vitiligo, especially in initiating repigmentation.
Introduction
Vitiligo is an acquired depigmentation disorder affecting about 1 percent of the population [1]. It usually affects younger patients and can be associated with great cosmetic and social problems. A large variety of therapeutic agents are being tried for the treatment of vitiligo, but an optimal treatment is not yet available. Recent studies reported an increased response rate for vitiligo patients treated with the combination of PUVA and calcipotriol [2, 3].
This study examines the efficacy of this combination in the treatment of vitiligo.
Methods
We enrolled 23 patients (16 females and 7 males) with essentially bilateral symmetrical lesions of vitiligo in a prospective trial over an 18-month period. The mean age of the patients was 36 years (range 19-73 yrs). Of these patients, 78 percent were classified as prototype IV or V. The median duration of disease was 11 years (range 1-31 yrs). All patients gave informed consent. The patients were advised to apply calcipotriol (0.005 %) twice daily over the right side of the body; the left side was not treated with calcipotriol. To prevent UVA inactivation, calcipotriol was applied after PUVA on phototherapy days.
An oral dose of 0.6 mg/kg 8-methoxypsoralen was given 2 hours before PUVA, 3 times per week. Waldman 7001K equipment was used as the UVA source. The PUVA was given to both sides while the calcipotriol was used only on the right side. The UVA treatment schedule was 0.5 J/cm² at the start of therapy. It was progressively increased to 5J/ cm². This dose maintains moderate-asymptomatic erythema in vitiliginous patches. To insure uniform treatment between patients, all of them were kept at this dose for the remainder of the sessions. Response to the treatment was evaluated clinically and by keeping photographic records at the start of therapy and all fifteen weeks. All patients received at least fortyfive sessions of PUVA.
Improvement was recorded as minimal (< 25 %); moderate (26-50 %); marked (> 50 %) or complete. The chi-square test was applied. All adverse events observed were recorded. Calcium and phosphorus serum levels were determined at the beginning and at the end of treatment.
Results
The 23 patients completed at least the fortyfive courses. For unresponsive patients (n = 5), the PUVA was then interrupted. All have lesions in the acral regions. At the fifteenth session, 16 patients (69 %) showed minimal to moderate improvement on calcipotriol-treated sides as compared to 12 patients (52 %) on nontreated sides (p = 0.015): Table 1.
|
|
No response | Minimal | Moderate | Marked |
| Calcipotriol + PUVA (n = 23) |
7 | 9 | 7 | 0 |
| PUVA (n = 23) |
11 | 12 | 0 | 0 |
|
|
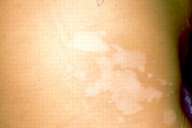
| Figure 1a | Figure 1b |
|---|---|
| Figures 1a and 1b. Bilateral lesions of vitiligo in a patient before treatment (a) right, (b) left. | |

|

|
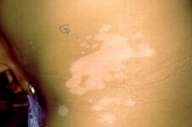
| Figure 2a | Figure 2b |
|---|---|
| Figures 2a and 2b. Repigmentation at the 15th session (a) in calcipotriol treated sides (b) in non-treated calcipotriol sides. Greater areas of repigmentation in (a). | |
|
|
No response | Minimal | Moderate | Marked |
| Calcipotriol + PUVA (n = 23) |
5 | 1 | 5 | 12 |
| PUVA (n = 23) |
5 | 2 | 9 | 7 |
At the forty-fifth session, 12 patients (52 %) showed marked improvement of the calcipotriol-treated side, whereas 7 patients (30 %) showed similar improvement of the PUVA-only side (p = 0.13): Table II. When repigmentation was complete, it was more intense in calcipotriol-treated areas. Mild irritation at the calcipotriol application site was reported by 2 patients (8.7 %). No significant difference was found between pre- and post-treatment levels of serum calcium and phosphorus.

|
|
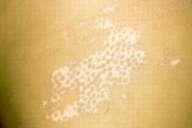
| Figure 3a | Figure 3b |
|---|---|
| Figures 3a and 3b. forty-fifth session: The patient showed repigmentation in the two sides (a, b). | |
|
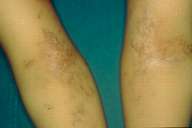
| Figure 4 |
|---|
| Figure 4. Complete repigmentation but more intense in the right side. |
Conclusions
Current treatment modalities for repigmenting vitiliginous macules are unsatisfactory [4]. Psoralen plus ultraviolet A (PUVA) appears to be the best method for achieving repigmentation [4], although variable results are reported [5]. Recent studies reported the efficacy of the combination of PUVA and topical calcipotriol [2, 3, 6, 7, 8].
Our study recorded a faster response in vitiligo lesions treated with PUVA and calcipotriol (p = 0.015) with more intense repigmentation in the calcipotriol treated side. However, lesions in acral regions did not respond to this therapy. The addition of topical calcipotriol to PUVA treatment was not associated with serious side effects.
Recent advances in the pathophysiology of vitiligo demonstrate defective calcium homeostasis in vitiliginous skin [9, 10].
Calcipotriol is a synthetic analogue of 1,25-dihydroxyvitamin D3. Most of the biological actions of calcipotriol and other vitamin D analogs are mediated by the vitamin D receptor [11, 12]. Many types of cells have vitamin D receptors, including keratinocytes, melanocytes, fibroblasts, and immune-system cells of the skin [6, 12]. The vitamin D receptor functions as a ligand-induced transcription factor, regulating the rate of gene expression involved in the control of calcium homeostasis and bone remodelling, control of hormone secretion, inhibition of cell growth, and induction of cell differentiation [13, 14, 15]. Calcipotriol is also an immunomodulator that interacts with cells, especially antigen-presenting cells [12, 16, 17, 18, 19]. The effect of calcipotriol on melanocytes may be direct or by means of these immunomodulatory actions.
Our study supports the efficacy of PUVA and topical calcipotriol, especially in initiating repigmentation. This combination may shorten the duration of UVA exposure, leading to decreases in the dose-dependent side effects related to PUVA [6].
References
1. Kovacs SO, Vitiligo. J Am Acad Dermatol. 1998 May;38(5 Pt 1):647-66. PubMed2. Parsad D, Saini R, Verma N. Combination of PUVAsol and topical calcipotriol in vitiligo. Dermatology. 1998;197(2):167-70. PubMed
3. Parsad D, Saini R, Nagpal R. Calcipotriol in vitiligo: a preliminary study. Pediatr Dermatol. 1999 Jul-Aug;16(4):317-20. PubMed
4. Jimbow K. Vitiligo. Therapeutic advances. Dermatol Clin. 1998 Apr;16(2):399-407. PubMed
5. McNeely W, Goa KL. 5-Methoxypsoralen. A review of its effects in psoriasis and vitiligo. Drugs. 1998 Oct;56(4):667-90. PubMed
6. Yalçin B, Sahin S, Bükülmez G, Karaduman A, Atakan N, Akan T, Kölemen F. Experience with calcipotriol as adjunctive treatment for vitiligo in patients who do not respond to PUVA alone: a preliminary study. J Am Acad Dermatol. 2001 Apr;44(4):634-7. PubMed
7. Ermis O, Alpsoy E, Cetin L, Yilmaz E. s the efficacy of psoralen plus ultraviolet A therapy for vitiligo enhanced by concurrent topical calcipotriol? A placebo-controlled double-blind study. Br J Dermatol. 2001 Sep;145(3):472-5. PubMed
8. Ameen M, Exarchou V, Chu AC. Topical calcipotriol as monotherapy and in combination with psoralen plus ultraviolet A in the treatment of vitiligo. Br J Dermatol. 2001 Sep;145(3):476-9. PubMed
9. Schallreuter KU, Pittelkow MR, Defective calcium uptake in keratinocyte cell cultures from vitiliginous skin. Arch Dermatol Res. 1988;280(3):137-9. PubMed
10. Schallreuter KU, Pittelkow MP, Swanson NN/ Defective calcium transport in vitiliginous melanocytes. Arch Dermatol Res. 1996;288(1):11-3. PubMed
11. Milde P, Hauser U, Simon T, Mall G, Ernst V, Haussler MR, Frosch P, Rauterberg EW. Expression of 1,25-dihydroxyvitamin D3 receptors in normal and psoriatic skin. J Invest Dermatol. 1991 Aug;97(2):230-9. PubMed
12. Guilhou JJ. Calcipotriol. Ann Dermatol Venereol. 2001 Mar;128(3 Pt 1):229-37. French. PubMed
13. Dusso AS, Brown AJ. Mechanism of vitamin D action and its regulation. Am J Kidney Dis. 1998 Oct;32(2 Suppl 2):S13-24. Review. PubMed
14. Fogh K, Kragballe K. Recent developments in vitamin D analogs. Curr Pharm Des. 2000 Jun;6(9):961-72. PubMed
15. Bikle DD, Ng D, Tu CL, Oda Y, Xie Z. Calcium- and vitamin D-regulated keratinocyte differentiation. Mol Cell Endocrinol. 2001 May 25;177(1-2):161-71. PubMed
16. van Etten E, Branisteanu DD, Verstuyf A, Waer M, Bouillon R, Mathieu C. Analogs of 1,25-dihydroxyvitamin D3 as dose-reducing agents for classical immunosuppressants. Transplantation. 2000 May 15;69(9):1932-42. PubMed
17. Abdel-Malek ZA, Ross R, Trinkle L, Swope V, Pike JW, Nordlund JJ. Hormonal effects of vitamin D3 on epidermal melanocytes. J Cell Physiol. 1988 Aug;136(2):273-80. PubMed
18. Bikle DD. Vitamin D: a calciotropic hormone regulating calcium-induced keratinocyte differentiation. J Am Acad Dermatol. 1997 Sep;37(3 Pt 2):S42-52. PubMed
19. Bagot M, Charue D, Lescs MC, Pamphile RP, Revuz J. Immunosuppressive effects of 1,25-dihydroxyvitamin D3 and its analogue calcipotriol on epidermal cells. Br J Dermatol. 1994 Apr;130(4):424-31. PubMed
© 2003 Dermatology Online Journal

