Unilateral laterothoracic exanthema with coincident evidence of Epstein Barr virus reactivation: Exploration of a possible link
Published Web Location
https://doi.org/10.5070/D31z1140k9Main Content
Unilateral laterothoracic exanthema with coincident evidence of Epstein Barr virus reactivation: Exploration of a possible
link
Noah Scheinfeld
Dermatology Online Journal 13 (3): 13
Columbia University, New York.Scheinfeld@earthlink.netAbstract
Unilateral laterothoracic exanthem (ULE) was first described in 1962 in the United States and comprehensively elaborated in 1992. Although ULE most commonly occurs in children, ULE can occur in adults. ULE may or may not be preceded by a viral prodrome and is marked by coalescing erythematous papules predominately on one side of the body. ULE usually lasts 4-6 weeks but can last as little as 2 weeks. It has inconsistently been linked to viral infection, in particular parvovirus B-19. I note ULE in an adult with concurrent reactivation of Epstein Barr virus (EBV) that lasted 4 weeks. The role of the reactivation of EBV in human disease and ULE is explored.
Unilateral laterothoracic exanthem (ULE) (also termed asymmetric periflexural exanthem of childhood) was first described in 1962 in the United States and comprehensively elaborated in 1992 [1, 2, 3, 4, 5, 6]. It usually manifests as unilateral erythema without systemic symptoms. ULE has been linked to viral infection, in particular parvovirus B-19. While ULE most commonly occurs in children, ULE can occur in adults.
Clinical synopsis
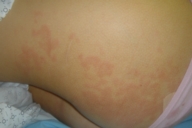 |
| Figure 1 |
|---|
A 35 year-old woman presented with an asymptomatic 2-week history of a unilateral erythematous eruption on her right flank (Fig. 1) without palpable lymph nodes. Skin biopsy and viral testing were performed. The skin biopsy showed a superficial and deep infiltrate of lymphocytes, and a lymphocytic infiltrate surrounding blood vessels and eccrine ducts (Figs. 2 and 3). Viral titers for demonstrated an EBV early antigen D Elisa Value (EV) of 81.9 (normal range 0-19.9), an EBV capsid antibody IgM EV of 2.8 (normal range 0-19.9), an EBV capsid antibody IgG EV of >20 (normal range 0-19.9) (positive), an EBV nuclear antigen EV of 87 (normal range 0-19.9) and a Parvovirus B-19 Index EV of <0.9 (normal range <0.9); results consistent with a EBV reactivation. Two weeks after the initial examination the eruption had almost abated altogether and four weeks later serologic testing revealed an EBV capsid antibody IgM EV of 2.8 (normal range 0-19.9), EBV capsid antibody IgG EV of >20 (normal range 0-19.9), EBV early antigen D EV of 2.8 (normal range 0-19.9), EBV nuclear antigen EV of 2.1 (normal range 0-19.9), and an parvovirus B-19 Index EV of <0.9 (normal range <0.9). Bacterial cultures and titers were negative at the time of examination and two weeks later.
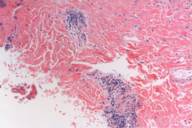 | 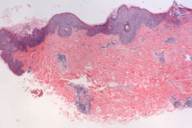 |
| Figure 2 | Figure 3 |
|---|
Discussion
The course of this case of ULE was typical. ULE may or may not be preceded by a viral prodrome and is marked by coalescing erythematous papules predominately on one side of the body. It occurs mostly in children with a mean age of 2 years of age but has been reported in adults. The largest series of cases of ULE of 48 children found a mean duration of 5 weeks. A more recent report describing a variant of ULE termed unilateral mediothoracic exanthem noted spontaneous remission was seen 2 and 3 weeks after rash onset in the child and the adult, respectively.
A viral cause of ULE has been postulated but inconsistently identified. The most virus most closely linked to ULE is parvovirus B-19 [2]. The histology of ULE has been noted to manifest consistently with a superficial perivascular infiltrate of lymphocytes that often forms a tight cuff around blood vessels and eccrine ducts and to manifest sometimes with miliarial spongiosis and exocytosis of lymphocytes into the acrosyringium [3, 4]. There does not have to be a rise in IgG or IgM levels during reactivation of EBV [5].
This case suggests that (1) EBV might be a cause ULE (2) Viral IgM might not necessarily elevated when a virus is the causal etiology of ULE (3) ULE may be linked to reactivation of a viral infection rather than acute viral infection and (4) ULE has a consistent clinical and histological presentation irrespective of causal virus.
The presence of IgG to EBV and detectable EBV virus which disappeared in short order while proof of viral reactivation is not itself EBV caused this unilateral eruption. The role of reactivation of viral infections (EBV but other herpes viruses as well) in skin eruptions and disease is an area that has gained increasing attention [8]. Whether the reactivation of virus found serologically in disease states is a causal phenomenon or an epiphenomenon is a controversial and complex matter. Quantities of EBV increase during times of stress that have been noted to include space flight, marathon training and antarctic exploration without evidence of disease. Thus the significance of increased levels of circulating EBV in otherwise healthy individuals is unclear. Increased and detectable viral replication when found while a disease state exists is suggestive that the EBV has a role in a disease not definitive proof. Complicating this calculation is that as will be discussed below, in certain pathologic states, multiple viruses can simultaneously increase their number while a disease state arises and progresses. Finally, the basis for a the clinical appearance of a particular eruption caused by EBV has yet to be defined.
Multiple techniques exist for assessing viral activity including the following: (1) polymerase chain reaction testing of blood fluid tissue for the presence of virons and viral DNA sequences; (2) immunohistological and immunofluorescent testing; and (3) viral cultures. The most means of testing that are positive, the more definite the role that reactivation can be ascribed to a pathologic state.
One well defined relationship of viral reactivation includes: the association of human herpes virus 6 infection with drug reaction with eosinophilia and systemic symptoms and anticonvulsant hypersensitivity syndrome [9]. This relationship has also been found with reactivation of cytomegalovirus (CMV) or EBV. Interestingly, when HHV-6 titers increase reactivation of HHV-7, CMV and/or EBV can also manifest following such drug eruptions [10]. In one study, the cascade of virus reactivation initiated by HHV-6 or EBV extended to EBV or HHV-7, and eventually to CMV [11].
Reactivation of Epstein-Barr virus has been linked to inflammatory diseases, diseases with inflammatory and neoplastic qualities, neoplasms, and immunosuppressive medications. The inflammatory diseases reactivation EBV has been lined to include the following: (1) ampicillin-induced cutaneous eruption associated with Epstein-Barr virus [12]; (2) florid reactions to mosquito bites [13]; (3) anticonvulsant hypersensitivity syndrome [14]; (4) Sjogren's syndrome [15, 16]; and (5) Gianotti-Crosti syndrome [17]. Although some reports link pityriasis lichenoides to EBV, I have not located any report linking it to reactivation of EBV. Diseases straddling inflammatory and neoplastic disease, such as reactive Epstein-Barr virus-related polyclonal lymphoproliferative disorder[18] and fatal hemophagocytic syndrome, have been linked to EBV reactivation [19]. The neoplastic diseases reactivation of EBV has been linked to include the following: Burkitt's lymphoma; Hodgkin's lymphoma; lymphomas and lymphoproliferative diseases in the immunocompromised; and nasopharyngeal and gastric carcinoma [20].
Reports linking immunosuppressive medications to diseases associated with reactivation of EBV include the following: (1) a patient who developed an EBV infection with the clinical features of an infectious mononucleosis, after immune suppression with cyclosporine and two courses of anti-thymocyte globulin for severe aplastic anemia [21]; (2) patients with lymphoma-related methotrexate use [22]; and (3) a patient taking prednisone preceding fatal fulminant hepatitis [23].
Conclusion
Much work still needs to done fitting the role of viruses into to human disease. This report suggests that EBV is related to ULE. Understanding the findings in this report will require other reports to explicate whether the relationship of ULE and EBV is causal or incidental. The basis for the unilateral appearance of this eruption is also in need of explication as it follows a pattern that no other eruption follows.
References
1. Bodemer C, de Prost Y. Unilateral laterothoracic exanthem in children: A new disease? J Am Acad Dermatol 1992;27:693-6. PubMed2. Pauluzzi P, Festini G, Gelmetti C. Asymmetric periflexural exanthem of childhood in an adult patient with parvovirus B19. J Eur Acad Dermatol Venereol. 2001;15:372-4. PubMed
3. Coustou D, Leaute-Labreze C, Bioulac-Sage P, Labbe L, Taieb A. Asymmetric periflexural exanthem of childhood: a clinical, pathologic, and epidemiologic prospective study. Arch Dermatol. 1999;135:799-803. PubMed
4. McCuaig CC, Russo P, Powell J, Pedneault L, Lebel P, Marcoux D. Unilateral laterothoracic exanthem. A clinicopathologic study of forty-eight patients. J Am Acad Dermatol. 1996;34:979-84. PubMed
5. Obel N, Hoier-Madsen M, Kangro H. Serological and clinical findings in patients with serological evidence of reactivated Epstein-Barr virus infection. APMIS. 1996;104:424-8. PubMed
6. Chuh AA, Chan HH. Unilateral mediothoracic exanthem: a variant of unilateral laterothoracic exanthem. Cutis. 2006 Jan;77(1):29-32. PubMed
7. Drago F, Rebora A. Viral reactivation and skin eruptions. Dermatology. 2003;207(1):1-2. PubMed
8. Lipsker D, Saurat JH. A new concept: paraviral eruptions. Dermatology. 2005;211(4):309-11. PubMed
9. Descamps V, Valance A, Edlinger C, Fillet AM, Grossin M, Lebrun-Vignes B, Belaich S, Crickx B. Association of human herpesvirus 6 infection with drug reaction with eosinophilia and systemic symptoms. Arch Dermatol. 2001 Mar;137(3):301-4. PubMed
10. Seishima M, Yamanaka S, Fujisawa T, Tohyama M, Hashimoto K. Reactivation of human herpesvirus (HHV) family members other than HHV-6 in drug-induced hypersensitivity syndrome. Br J Dermatol. 2006 Aug;155(2):344-9. PubMed
11. Kano Y, Hiraharas K, Sakuma K, Shiohara T. Several herpesviruses can reactivate in a severe drug-induced multiorgan reaction in the same sequential order as in graft-versus-host disease. Br J Dermatol. 2006 Aug;155(2):301-6. PubMed
12. Saito-Katsuragi M, Asada H, Yokoi S, Niizeki H, Miyagawa S. Ampicillin-induced cutaneous eruption associated with Epstein-Barr virus reactivation. J Am Acad Dermatol. 2005 May;52(5 Suppl 1):S127-8. PubMed
13. Shigekiyo T, Ohmori H, Chohraku M, Ohtsuka S, Yamabe K, Takishita Y, Takai S, Takahashi M, Wakatsuki S. Unusual skin reactions after mosquito bites and Epstein-Barr virus reactivation in a patient with mantle cell lymphoma.Intern Med. 2004 Oct;43(10):986-9. PubMed
14. Chang JY, Kim SC. Anticonvulsant hypersensitivity syndrome associated with epstein-barr virus reactivation. Yonsei Med J. 2007 Apr 30;48(2):317-20. PubMed
15. Fox RI, Luppi M, Kang HI, Pisa P. Reactivation of Epstein-Barr virus in Sjogren's syndrome. Springer Semin Immunopathol. 1991;13(2):217-31. PubMed
16. Miyasaka N, Saito I, Haruta J. Possible involvement of Epstein-Barr virus in the pathogenesis of Sjogren's syndrome. Clin Immunol Immunopathol. 1994 Aug;72(2):166-70. PubMed
17. Terasaki K, Koura S, Tachikura T, Kanzaki T. Gianotti-Crosti syndrome associated with endogenous reactivation of Epstein-Barr virus. Dermatology. 2003;207(1):68-71. PubMed
18. Martin SI, Zukerberg L, Robbins GK. Reactive Epstein-Barr virus-related polyclonal lymphoproliferative disorder in a patient with AIDS.Clin Infect Dis. 2005 Oct 15;41(8):e76-9. PubMed
19. Hasselblom S, Linde A, Ridell B. Hodgkin's lymphoma, Epstein-Barr virus reactivation and fatal haemophagocytic syndrome. J Intern Med. 2004 Feb;255(2):289-95. PubMed
20. Pattle SB, Farrell PJ. The role of Epstein-Barr virus in cancer. Expert Opin Biol Ther. 2006 Nov;6(11):1193-205. PubMed
21. Calistri E, Tiribelli M, Battista M, Michelutti A, Corbellino M, Viale P, Fanin R, Damiani D. Epstein-Barr virus reactivation in a patient treated with anti-thymocyte globulin for severe aplastic anemia.Am J Hematol. 2006 May;81(5):355-7. PubMed
22. Feng WH, Cohen JI, Fischer S, Li L, Sneller M, Goldbach-Mansky R, Raab-Traub N, Delecluse HJ, Kenney SC. Reactivation of latent Epstein-Barr virus by methotrexate: a potential contributor to methotrexate-associated lymphomas. J Natl Cancer Inst. 2004 Nov 17;96(22):1691-702. PubMed
23. Cacopardo B, Nunnari G, Mughini MT, Tosto S, Benanti F, Nigro L. Fatal hepatitis during Epstein-Barr virus reactivation. Eur Rev Med Pharmacol Sci. 2003 Jul-Aug;7(4):107-9. PubMed
© 2007 Dermatology Online Journal

