Cutaneous Lichenoid dermatitis associated with imatinib mesylate
Published Web Location
https://doi.org/10.5070/D31jg6p60zMain Content
Chung-Yin Stanley Chan MD, John Browning MD, Megan J Smith-Zagone MD, Paul T Martinelli MD, Sylvia Hsu MD
Dermatology Online Journal 13 (2): 29
Department of Dermatology, Baylor College of Medicine, Houston, TexasImatinib mesylate (STI 571, Gleevec®) is a specific molecular inhibitor of the fusion protein bcr-abl, a constitutively active tyrosine kinase [1]. It was the first approved drug to directly inhibit the signal of a protein known to cause a cancer, commonly used in the treatment of chronic myelogenous leukemia (CML) and gastrointestinal stromal tumors (GIST). Skin reactions resulting from Gleevec® have been well-described, however, reports of an associated lichenoid dermatitis are exceedingly rare. There have been only four case reports of lichenoid reactions associated with Gleevec®, two with oral involvement only [2, 3], and two with only cutaneous manifestations including one without histologic confirmation [4, 5]. We report a rare case of cutaneous lichenoid dermatitis associated with Gleevec®.
A 56-year-old male with a 3-year history of chronic myelogenous leukemia presented with pruritic, violaceous papules and plaques over his legs, thighs, chest and arms for one month duration. The patient had no mucocutaneous lesions. He had been treated with Gleevec® 600 mg daily for 3 months before onset of the rash. Prior to evaluation by dermatology, the patient was given triamcinolone 0.1% cream topically and prednisone 20 mg by mouth for five days, with moderate improvement in the lesions. The only other medications the patient was taking were promethazine and lansoprazole. On physical examination, there were scattered pruritic, violaceous, scaly papules on the trunk and extremities.
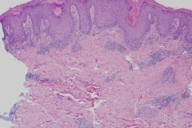
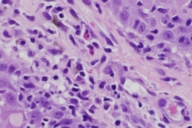
Histopathologic examination of a 4 mm punch biopsy taken from a papule on the left thigh revealed a lichenoid band of lymphocytes and histiocytes with numerous eosinophils. There was also hyperkeratosis, focal parakeratosis, acanthosis and a superficial and deep perivascular lymphovascular infiltrate (Figs. 1 and 2).
The histologic pattern of our biopsy is highly consistent with a lichenoid drug reaction. Although this pattern can be indistinguishable from idiopathic lichen planus, supportive histologic characteristics for a lichenoid drug eruption include focal parakeratosis, the presence of eosinophils, exocytosis of lymphocytes into the upper epidermis and a deep perivascular infiltrate [6]. Furthermore, the patient's onset of the eruption after 3 months correlates well with the timeframe of previous reports. Finally, the patient subsequently stopped Gleevec® for 3 months with improvement in the lesions and upon reinitiating, the lesions recurred.
A plethora of drugs have been associated with lichenoid eruptions [7]. Innumerable soldiers in World War II experienced lichenoid eruptions secondary to malarial prophylaxis with mepacrine [8]. Lichenoid eruptions have also been reported with demeclocycline, thiazide diuretics, streptomycin, isoniazid, metropromazine, levopromazine, methyldopa, pyrimethenamine [9], beta blockers [10. 11], angiotensin-converting enzyme inhibitors [12, 13] , and naproxen [14]. Many other medications have also been associated with lichenoid reactions.
Non-lichenoid cutaneous reactions secondary to Gleevec® have been well described and are the most common non-hematologic adverse events associated with its use [15]. The most common cutaneous reactions include morbilliform eruptions and cutaneous edema, particularly, periorbital edema [16]. Cases of severe generalized skin eruptions such as erythema multiforme [17], acute generalized exanthemous pustulosis [18], and toxic epidermal necrolysis [19] have also been reported with Gleevec® use. Cutaneous reactions to Gleevec® appear to be dose related, appearing more frequently and severely in patients on doses of 600 mg daily or greater [20], as in our patient. With an increasing number of patients with being treated with Gleevec®, we anticipate further reports of lichenoid and other cutaneous reactions associated with its use.
References
1. Mauro MJ, Deininger MW. Chronic myeloid leukemia in 2006: a perspective. Haematologica. 2006 Feb;91(2):152 PubMed.2. Lim DS, Muir J. Oral lichenoid reaction to imatinib (STI 571, Gleevec). Dermatology. 2002;205(2):169-71 PubMed.
3. Ena P, Chiarolini F, Siddi GM, Cossu A. Oral lichenoid eruption secondary to imatinib (Glivec).J Dermatolog Treat. 2004 Jul;15(4):253-5 PubMed.
4. Lichenoid cutaneous reaction to imatinib. Ann Dermatol Venereol. 2004 Jun-Jul;131(6-7 Pt 1):571-3. [abstract only] PubMed.
5. Prabhash K, Doval DC. Lichenoid eruption due to imatinib. Indian J Dermatol Venereol Leprol. 2005 Jul-Aug;71(4):287-8 PubMed.
6. Van Den Haute V, Antoine JL, Lachapelle JM. Histopathological discriminant criteria between lichenoid drug eruption and idiopathic lichen planus: retrospective study on selected samples. Dermatologica. 1989;179(1):10-3 PubMed.
7. Black MM. Lichen planus and lichenoid disorders. In: Champion RH, Burton JL, Burns DA, editors. Textbook of Dermatology. 6th ed. Oxford, England: Blackwell Science, 1998: 1916-8.
8. Bauer F. Quinacrine hydrochloride drug eruption (topical lichenoid dermatitis). J Am Acad Dermatol 1981; 4: 239-48 PubMed.
9. Cutler TP. Lichen planus caused by pyrimethamine. Clin Exp Dermatol 1980; 5:253-6 PubMed.
10. Hawk JLM. Lichenoid drug eruption induced by propanolol. Clin Exp Dermatol 1980; 5:93-6 PubMed
11. Gange RW, Jones EW. Bullous lichen planus caused by labetalol. Br Med J 1978; 1:816-17 PubMed
12. Roter SV, Mainetti C. Donath R et al. Enalapril induced lichen planus-like eruption. J Am Acad Dermatol 1995; 32 :293-5 PubMed.
13. Philips WG, Vaughan-Jones S, Jenkins R et al. Captopril-induced lichenoid eruption. Clin Exp Dermatol 1994; 19: 317-20 PubMed.
14. Heymann WR, Lerman JS, Luftshein S. Lichen Naproxen-induced lichen planus. J Am Acad Dermatol. 1984Feb; 10 (Z Pt1): 299-301 PubMed.
15. O'Brien SG, Guilhot F, Larson RA, Gathmann I, Baccarani M, Cervantes F, 16. Cornelissen JJ, Fischer T, Hochhaus A, Hughes T, Lechner K, Nielsen JL, Rousselot P, Reiffers J, Saglio G, Shepherd J, Simonsson B, Gratwohl A, Goldman JM, Kantarjian H, Taylor K, Verhoef G, Bolton AE, Capdeville R, Druker BJ; Imatinib compared with interferon and low-dose cytarabine for newly diagnosed chronic-phase chronic myeloid leukemia. N Engl J Med. 2003 Mar 13;348(11):994-1004 PubMed.
16. Scheinfeld N. Imatinib mesylate and dermatology part 2: a review of the cutaneous side effects of imatinib mesylate. J Drugs Dermatol. 2006 Mar;5(3):228-31 PubMed.
17. Hsiao LT, Chung HM, Lin JT, et al. Stevens-Johnson syndrome after treatment with STI571: a case report. Br J Haematol. 2002;117:620-622 PubMed.
18. Brouard MC, Prins C, Mach-Pascual S, Saurat JH. Acute generalized exanthematous pustulosis associated with STI571 in a patient with chronic myeloid leukemia. Dermatology. 2001;203(1) 57-9 PMIN: 11549802.
19. Schaich M, Schakel K, Illmer T, Ehninger G, Bornhauser M. Severe epidermal necrolysis after treatment with imatinib and consecutive allogeneic hematopoietic stem cell transplantation. Ann Hematol. 2003;82:303-4 PubMed.
20. Brouard M, Saurat JH. Cutaneous reactions to STI571. N Engl J Med. 2001 Aug 23;345(8):618-9 PubMed.
© 2007 Dermatology Online Journal