Candida glabrata infection in gastric carcinoma patient mimicking cutaneous histoplasmosis
Published Web Location
https://doi.org/10.5070/D31cx1g3tvMain Content
Candida glabrata infection in gastric carcinoma patient mimicking cutaneous histoplasmosis
Dijana Gugic MD, Timothy Cleary MD, Vladimir Vincek MD PhD
Dermatology Online Journal 14 (2): 15
Department of Pathology, University of Miami, Jackson Memorial Hospital. dijana.gugic@st.t-com.hrAbstract
Candida glabrata is the second most common Candida species detected among hospitalized patients in USA. In tissue C. glabrata present as yeasts, 3-5 microns in size, which are difficult to visualize on H&E stained slides but can be detected on Grocott methenamine silver (GMS) stained slides. The presence of yeasts only, without any hyphal elements, makes C. glabrata difficult to distinguish from Histoplasma capsulatum yeasts that are of similar size. Mycology culture is the method of choice for definitive identification of C. glabrata. Rapid identification is necessary, as mortality rate due to C. glabrata infection in immunocompromised patients is particularly high. We herein report a patient with inoperable gastric carcinoma, who developed cutaneous and septic form of C. glabrata infection.
Clinical synopsis
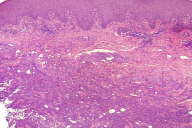 |
| Figure 1 |
|---|
| Figure 1. H&E stained skin punch biopsy showing deep dermal and subcutaneous necrosis and acute inflammation (40X). |
We present the case of 62 year-old Haitian man diagnosed with infiltrating, poorly differentiated gastric carcinoma. The tumor was found to be inoperable and patient was given palliative therapy. At the time of diagnosis, the patient had a fever of 39.5°C. On the lateral side of his right arm, an indurate, pale tan plaque, measuring 0.8 cm in largest diameter, was seen. A punch biopsy of skin lesion was obtained for histological evaluation as well as for bacterial, mycobacterial and fungal cultures. Histological examination showed deep dermal and subcutaneous necrosis with acute neutrophilic inflammation. No infectious organisms could be readily appreciated on routine hematoxylin-eosin (H&E) stained slides (Figs. 1 and 2). However, the GMS revealed numerous yeasts, 3-5 microns in size, present within the dermis and subcutis (Fig. 3). Because only yeasts without hyphal and pseudohyphal elements were found, histoplasmosis was suspected. Mycology culture was recommended to further classify the fungal elements. Candida glabrata was identified in the culture whilst no cultures were positive for Histoplasma spp. At the same time C. glabrata and Klebsiella pneumoniae were found in the blood culture. The patient was treated with caspofungin (50 mg IV) and ceftriaxone (2 mg IV). Given the persistent infection that did not improve despite the administered drugs, he was not started on chemotherapy. A month after initial diagnosis the patient expired.
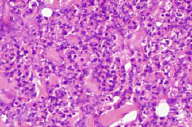 | 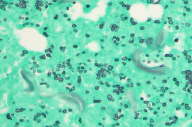 |
| Figure 2 | Figure 3 |
|---|---|
| Figure 2. H&E stained skin punch biopsy showing deep necrosis and neutrophilic inflammation (200X). Figure 3. GMS stained skin punch biopsy demonstrating fungal spores of C. glabrata. | |
Discussion:
The risk of opportunistic infections is greatly increased in patients who are immunocompromised because of AIDS, cancer, chemotherapy and organ or bone-marrow transplantation [1, 2]. In the last few decades the importance of candidemia in oncology patients, its changing epidemiology patterns, and significant morbidity and mortality rate has been recognized [3-18]. In humans, candidiasis is caused by 13 species, most frequently by the following: C. albicans, C. tropicalis, C. glabrata, C. krusei, C. parapsilosis, C. lusitanie and others. Historically, C. albicans accounted for over half of the isolates recovered from infected patients. However, more recent epidemiological data reveal a mycological shift from C. albicans to the non-albicans Candida spp, especially C. glabrata and C. krusei [3-8, 19, 20]. The results of the 2003 longitudinal SENTRY antimicrobial surveillance program positioned C. glabrata, with 12.7 percent of isolates, as the second ranked among Candida species in patients from North America. Most of C. glabrata infections arise from the host's endogenous microflora [2]. During a prospective evaluation of Candida species associated with either clinically significant colonization or infection, 26.9 percent of C. glabrata isolates showed in vitro resistance to fluconazole (MIC of > or = 64 μg/ml) and reduced susceptibility to newer azoles (ravuconazole, voriconazole) [12, 21].
Clinically, candidiasis may present as acute and chronic mucocutaneous form, and as disseminated disease [22]. C. albicans can cause localized and disseminated disease whereas other Candida species are associated with systemic disease more commonly then localized disease. Histologically, mucocutaneous candidiasis has following features: 3-6μm yeasts, pseudohyphae and hyphae in stratum corneum, subcorneal pustules, and vesicles [23]. Occasionally, candidal granuloma may form with prominent hyperkeratosis and papillomatosis, together with dense dermal infiltrate of giant cells. Disseminated candidiasis, on the other hand, demonstrates pseudohyphae, hyphae, and yeasts (which may show budding) localized in dermis near blood vessels with perivascular inflammatory infiltrate and leukocytoclastic vasculitis [23]. In contrast to C. albicans tissue, infection with C. glabrata present as yeasts 3-5 microns in size that are difficult to visualize on H&E stained slides but can be detected on GMS or PAS stained slides. The presence of yeasts only, without any hyphal elements, makes C. glabrata difficult to distinguish from Histoplasma capsulatum yeasts that are just slightly smaller (2-4 microns).
Patients with solid tumors (such was in our case), with acute leukemia, and hematological patients on antifungal prophylaxis are significantly associated with non-albicans candidemia. The reported overall 30-day mortality is 39 percent. In a univariate analysis, C. glabrata is associated with the highest mortality rate (odds ratio 2.6) [10].
Dermatopathologists may not be familiar with the histological presentation of C. glabrata. This case highlights that for immunocompromised cancer patients presenting with cutaneous lesions with histomorphology resembling H. capsulatum infection, one should always take into consideration the following: first (i) the possible presence of underlying life-threatening systemic mycosis and second (ii) Candida glabrata may be the infectious agent (especially in the light of new epidemiology trends which indicate increased frequency of mucosal and systemic infections caused by this agent). Early and accurate classification of fungal elements should secure a choice of an efficient antimycotic drug and, ultimately, a higher survival rate for the patient. Mycology culture is still the gold standard for identification of fungi, especially species that do not form pseudohyphae in vivo.
References
1. Hoffman HL, Ernst EJ, Klepser ME. Novel triazole antifungal agents. Expert Opin Investig Drugs. 2000;9(3):593-605. PubMed2. Kao AS, Brandt ME, Pruitt WR, Conn LA, Perkins BA, Stephens DS, Baughman WS, Reingold AL, Rothrock GA, Pfaller MA, Pinner RW, Hajjeh RA. The epidemiology of candidemia in two United States cities: results of a population-based active surveillance. Clin Infect Dis. 1999 Nov;29(5):1164-70. PubMed
3. Fidel PJ, Vazquez, J. and Sobel J. Candida glabrata: Review of epidemiology, pathogenesis, and clinical disease with comparison to C. albicans. Clin Microbiol Rev. 1999;00:80-96. PubMed
4. Beck-Sague, CM, and TR Jarvis. Secular trends in the epidemiology of nosocomial fungal infections in the United States. J Infect Dis. 1993;167:1247-1251, PubMed
5. Komishian, SV, AK Uwaydah, and JD Sobel. Fungemia caused by Candida species and Toluropsis glabrata in the hospitalized patient: frequency, characteristics, and evaluation of factors influencing outcome. Rev Infect Dis. 1989;11:379-390. PubMed
6. Merz, WG, JE Karp, D Schron, and R. Saral. Increased incidence of fungemia caused by Candida krusei. J. Clin. Microbiol. 1972;24:581-584. PubMed
7. Abi-Said D, Anaissie E, Uzun O, Raad I, Pinczowski H, Vartivarian S. The epidemiology of hematogenous candidiasis caused by different Candida species. Clin Infect Dis. 1997;24(6):1122-8. PubMed
8. Redding SW, Dahiya MC, Kirkpatrick WR, Coco BJ, Patterson TF, Fothergill AW, Rinaldi MG, Thomas CR Jr. Candida glabrata is an emerging cause of oropharyngeal candidiasis in patients receiving radiation for head and neck cancer. Oral Surg Oral Med Oral Pathol Oral Radio Endod. 2004;97(1):47-52. PubMed
9. Torres HA, Kontoyiannis DP, Rolston KV. High-dose fluconazole therapy for cancer patients with solid tumors and candidemia: an observational, noncomparative retrospective study. Support Care Cancer. 2004 Jul;12(7):511-6. PubMed
10. Viscoli C, Girmenia C, Marinus A, Collette L, Martino P, Vandercam B, Doyen C, Lebeau B, Spence D, Krcmery V, De Pauw B, Meunier F. Candidemia in cancer patients: a prospective, multicenter surveillance study by the Invasive Fungal Infection Group (IFIG) of the European Organization for Research and Treatment of Cancer (EORTC). Clin Infect Dis. 1999 May;28(5):1071-9. PubMed
11. Wingard JR. Importance of Candida species other than C. albicans as pathogens in oncology patients. Clin Infect Dis. 1995 Jan;20(1):115-25. PubMed
12. Safdar A, Armstrong D, Cross EW, Perlin DS. Prospective epidemiologic analysis of triazole-resistant nosocomial Candida glabrata isolated from patients at a comprehensive cancer center. Int J Infect Dis. 2002 Sep;6(3):198-201. PubMed
13. Antoniadou A, Torres HA, Lewis RE, Thornby J, Bodey GP, Tarrand JP, Han XY, Rolston KV, Safdar A, Raad II, Kontoyiannis DP. Candidemia in a tertiary care cancer center: in vitro susceptibility and its association with outcome of initial antifungal therapy. Medicine (Baltimore). 2003 Sep;82(5):309-21. PubMed
14. Kontoyiannis DP, Reddy BT, Hanna H, Bodey GP, Tarrand J, Raad II. Breakthrough candidemia in patients with cancer differs from de novo candidemia in host factors and Candida species but not intensity. Infect Control Hosp Epidemiol. 2002 Sep;23(9):542-5. PubMed
15. Bodey GP, Mardani M, Hanna HA, Boktour M, Abbas J, Girgaway E, Hachem RY, Kontoyiannis DP, Raad II. The epidemiology of Candida glabrata and Candida albicans fungemia in immunocompromised patients with cancer. Am J Med. 2002;112:380-385. PubMed
16. De Marie S. New developments in the diagnosis and management of invasive fungal infections. Haematologica. 2000;85:88-93. PubMed
17. Richardson MD. Changing patterns and trends in systemic fungal infections. J Antimicrob Chemother. 2005;56(S1):5-11. PubMed
18. Safdar A, Chaturvedi V, Koll BS, Larone DH, Perlin DS, Armstrong D. Prospective, multicenter surveillance of Candida glabrata: fluconazole and itraconazole susceptibility profiles in bloodstream, invasive, and colonizing strains and differences between isolates from three urban teaching hospitals in New York City (Candida susceptibility trends study, 1998-1999). Antimicrob Agents Chemother. 2002 Oct;46(10):3268-3272. PubMed
19. Fairchild KD, Tomkoria S, Sharp EC, Mena FV. Neonatal Candida glabrata sepsis: clinical and laboratory features compared with other Candida species. Padiatr Infect Dis J. 2002;21(1):39-43. PubMed
20. Fuchs J, Mildbrat R, Pecher SA. Lobomycosis (keloidal blastomicosis): Case reports and overview. Cutis. 1990;46:227-234. PubMed
21. Messer SA, Jones RN, Fritsche TR. International Surveillance of Candida spp. and Aspergillus spp.: Report from the SENTRY antimicrobial surveillance program (2003). J Clin Microbiol. 2006;44(5):1782-1787. PubMed
22. Maize JC, Lynch PJ: Chronic mucocutaneous candidiasis of the adult. Arch Dermatol. 1972;105:96-98. PubMed
23. Barnhill RL. Textbook of Dermatopathology, 2nd edition, McGraw-Hill, New York, 2004.
© 2008 Dermatology Online Journal

