Frostbite injury of the foot from portable fire extinguisher
Published Web Location
https://doi.org/10.5070/D317r2w1z9Main Content
Frostbite injury of the foot from portable fire extinguisher
Celalettin Sever MD, Yalcin Kulahci MD, Fatih Uygur MD, Cihan Sahin MD
Dermatology Online Journal 15 (9): 10
Department of Plastic and Reconstructive Surgery and Burn Unit, Gülhane Military Medical Academy and Medical Faculty, Haydarpasa
Training Hospital, Istanbul, Turkey. drcsever@hotmail.comAbstract
Frostbite burns are uncommon and their etiologies are varied. We present a case of sudden frostbite burn of the left foot caused by carbon dioxide. The circumstances of this injury and preventive measures are discussed.
Introduction
Carbon dioxide is a clear, colorless, nonflammable gas. Although carbon dioxide mainly consists in the gaseous form, it also has a solid and a liquid form. Carbon dioxide is used widely for industrial purposes. The most familiar example is its use in soft drinks and beer, to make them fizzy [1, 2]. In addition, some fire extinguishers use carbon dioxide because it is denser than air. Because of this, carbon dioxide may blanket a fire.. It prevents oxygen from getting to the fire and as a result, the burning material is extinguished.
Solid carbon dioxide is always below -78°C at regular atmospheric pressure, regardless of the air temperature. Carbon dioxide gas in a steel cylinder, such as a fire extinguisher, is kept in pressurized liquid form. When the container is opened, the carbon dioxide expands to form a gas in the atmosphere.
Contact with the solid carbon dioxide gas for more than a second or two without proper protection may cause skin injury because of rapid and profound cooling, leading to localized cold injury and cellular destruction. Numbness develops because of inactivation of nerve sensation. Frostbite following exposure to cold gases is an occupational hazard [3, 4]. Despite its widespread use, carbon dioxide frostbite cases have not yet been reported in the literature.
Case report
A 20-year-old man of good general health was admitted to our burn care unit with frostbite involving the left foot. He told us that the frostbite injury resulted from contact with the fire extinguisher while he was playing football. The patient sprained his ankle and complained of severe pain. His teammates came to his rescue witha cooling spray, from a fire extinguisher. The carbon dioxide gas spray was sustained for a period of 30-45 seconds. Immediately after the exposure, the skin of his foot had gone white and cold. Afterwards, he initially felt numbness, but then increasing foot pain.
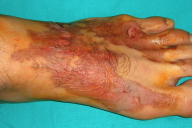 |
| Figure 1 |
|---|
Upon arrival at our hospital, 2 hours later, the patient was immediately taken to a cleaning tank. His foot was irrigated with 37°C heated and sterilized water for 15 minutes. The initial physical examination demonstrated second degree frostbite of the left foot (Fig. 1). The capillary circulation of his toes was clinically adequate. And all the toes were well perfused. Neurological examination showed no motor or sensory deficits. The remainder of the skin examination was normal.
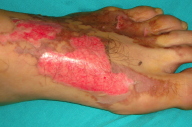 | 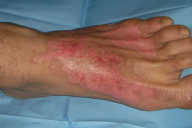 |
| Figure 2 | Figure 3 |
|---|
The patient was hospitalized immediately for close monitoring of his perfusion. He was given antiedema treatment, extremity elevation and intravenous fluids with high molecular weight dextran in saline (35ml/h). After 24 hours when the dorsum of the left foot was inspected, blisters were present. All hemorrhagic blisters were punctured and debrided under sedation (Fig. 2). Burn areas were dressed every day and healed gradually over two weeks (Fig. 3).
Discussion
Joseph Black, a Scottish chemist and physician, first identified carbon dioxide in the 1750s. At room temperatures (20-25°C), carbon dioxide is an odorless, colorless gas, which is faintly acidic and non-flammable. Carbon dioxide is one of the most abundant gasses in the atmosphere. Although carbon dioxide mainly consists in the gaseous form, it also has a solid and a liquid form. It may only be solid when temperatures are below -78°C. Liquid carbon dioxide mainly exists when carbon dioxide is dissolved in water. Carbon dioxide is only water-soluble when pressure is maintained. After the pressure drops it will form a mass of bubbles in the water.
Generally, the primary health dangers of carbon dioxide gas are: asphyxiation, kidney damage or coma, and frostbite. Asphyxiation is caused by the release of carbon dioxide in a confined or unventilated area. This can lower the concentration of oxygen to a level that is immediately dangerous for health. Kidney damage or coma is caused by a disturbance in the chemical equilibrium of the carbonate buffer. When carbon dioxide concentrations increase or decrease, causing the equilibrium to be disturbed, a life threatening situation may occur. In addition, contact with solid carbon dioxide for more than a second or two without proper protection may cause serious blisters and frostbite.
There are different types or classes of fire extinguishers, each of which extinguishes specific types of fire. Carbon dioxide extinguishers are most effective on liquid and electrical fires. Since the gas disperses quickly, these extinguishers are only effective from 3 to 8 feet. Carbon dioxide is heavier than oxygen so these extinguishers work by displacing or taking away oxygen from the surrounding area. The carbon dioxide is stored as a compressed liquid in the extinguisher; as it expands, it cools the surrounding air. Thus, it may cause frostbite, but this is very uncommon.
The extent of injury caused by carbon dioxide gas is determined by the surface area of exposed tissue, the volume of carbon dioxide gas that contacts the skin, and the duration of exposure time. Damage from acute exposure to carbon dioxide gas occurs within seconds. However there is some controversyl about the duration of exposure time that is required to causesevere tissue damage [5].The appearance of the superficial tissue is often an unreliable indicator of the viability of the underlying tissue, as is seen with other forms of burns [6]. The injury may be more severe than that caused by a thermal burn because the agent rapidly and deeply penetrates through the skin.
There is little information available in the medical literature on emergency treatments for carbon dioxide gas frostbite injuries. The first treatment step is to remove the patient from danger and minimize the duration of exposure. Unlike the thermal burn, the agent will continue to "burn" until the substance is inactivated or removed. In the first aid treatment, the carbon dioxide frost contaminated skin must be promptly washed using soap or mild detergent and water. Contaminated clothing must be removed immediately. For frostbite, immediate rewarming in a water bath between 42°C is recommended [3, 7].
In this case, the njury was superficial. The underlying tissues were healthy. Frostbite areas were dressed every day; capillary circulation was monitored.capillary circulation. If the duration of exposure had beenlonger, the deep tissues and muscles also could have been injured.
Prevention is the best strategy for reducing the morbidity and mortality of frostbite. The first step is increasing the awareness of the public and health care professionals of the risk of these injuries. Moreover, most of these injuries may be prevented by taking adequate precautions.
In conclusion, even short contact with carbon dioxide gas can result in severe frostbite injury. Decreasing the exposure time is an important first step in the treatment approach. After exposure, quick delivery of the patient to a burn center, is essential. Measures to prevent severe edema are important to avoid deep tissue damage.
References
1. Sutjiadi-Sia Y, Jaeger P, Eggers R. Interfacial tension of solid materials against dense carbon dioxide. J Colloid Interface Sci. 2008;320(1):268-74. [PubMed]2. Morita S, Iizuka S, Shibata M, Isotoshi Y, Sadaki I. Respiratory failure by inhalation of a fire extinguisher. J Trauma. 2005, 59(2):504. [PubMed]
3. Sever C, Ulkur E, Uygur F, Celikoz B. Hand burn caused by Freon gas. Burns. 2008; 34(8):1210-2. [PubMed]
4. Occupational Safety and Health Administration. Portable fire extinguishers (OSHA standard 1910.120). Washington, DC: US Department of Labor.
5. Hicks LM, Hunt JL, Baxter CR. Liquid propane cold injury: clinicopathologic and experimental study. J Trauma. 1979;19(9):701-3. [PubMed]
6. Aygit AC, Sarikaya A. Imaging of frostbite injury by technetium- 99m-sestamibi scintigraphy: a case report. Foot Ankle Int. 2002;23(1):56-9. [PubMed]
7. Heggers JP, McCauley RL, Phillips LG, Robson MC. Cold-induced injury: frostbite. In: Herndon DN, ed. Total burn care. London: WB Saunders; 1996;408-414.
© 2009 Dermatology Online Journal

