Chronic actinic dermatitis
Published Web Location
https://doi.org/10.5070/D317c4s0dvMain Content
Chronic actinic dermatitis
Alexandria V Booth MD, Stephanie Mengden MD, Nicholas A Soter MD, David Cohen MD, MPH
Dermatology Online Journal 14 (5): 25
Department of Dermatology, New York UniversityAbstract
A 71-year-old man presented with a six-year history of a pruritic, erythematous, blistering eruption of the face, chest, and arms. Clinical findings, histopathologic features, and phototests were consistent with a diagnosis of chronic actinic dermatitis. The patient also had contact allergy and photocontact allergy to multiple allergens. A discussion of chronic actinic dermatitis is presented.
Clinical synopsis
A 71-year-old Albanian man was referred to the Charles C. Harris Skin and Cancer Pavilion Allergy/Contact Dermatitis Section with a 6-year history of an intermittently pruritic, paroxysmally flaring eruption over the face, chest, arms, and legs. It typically begins as pruritus and edema with subsequent blisters and desquamation over a 3-week period. It initially affected his face but began to involve his entire body 4 years later. He felt that the eruption was worse after sun exposure and during the Spring. He noted temporary improvement with the use of systemic glucocorticoids. Past medical history included allergic rhinitis, cervical spondylitis, arthritis, and hypercholesterolemia. His only medication is diphenhydramine for pruritus. Allergy history included contact allergy to iodine, grass, and synthetic clothing as a child. He is a retired otolaryngologist, and his hobbies include watercolor painting, walking, and working on his computer.
Physical Examination
Confluent erythema was present on the face and V-area of the upper chest with sparing below the chin. Erythema with scattered erosions and lichenification was noted over the dorsal aspects of the arms and hands, with demarcation at the mid-portion of the upper arms and sparing of the proximal areas.
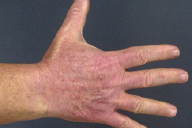 | 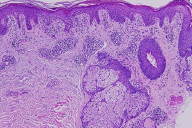 |
| Figure 1 | Figure 2 |
|---|
A complete blood count with differential analysis and comprehensive metabolic panel were normal. Antinuclear, anti-Ro, and anti-La antibodies were negative.
Phototests showed no immediate response to ultraviolet A, ultraviolet B or visible light; MEDa was low at 15 J/cm2, and MEDb was low at 5mJ/cm2.
Photopatch test reactions were positive to promethazine and chlorpromazine. Patch test reactions were positive to formaldehyde, quaternium-15, imidazolidinyl urea, diazolidinyl urea, ethyleneurea melamine formaldehyde, thiuram mix, carba mix, propylene glycol, methylchloroisothiazolone methylisothiazolone, and methylbibromoglutoaronitrile.
Histopathology
There is mild acanthosis, focal parakeratosis, and a few, necrotic keratinocytes within a spongiotic epidermis. Within the superficial and mid-to-deep dermis, there is a perivascular and periadnexal predominantly lymphocytic infiltrate with some exocytosis of lymphocytes. The inflammatory infiltrate is also composed of a few neutrophils and macrophages containing melanin.
Comment
Chronic actinic dermatitis (CAD) is the term first proposed in 1979 by Hawk and Magnus [1] to unify the conditions known as persistent light reactivity described by Wilkinson [2] in 1961, actinic reticuloid described by Ive et al. [3] in 1969, photosensitive eczema described by Ramsay and Black [4] in 1973, and photosensitivity dermatitis described by Frain-Bell et al. [5] in 1974. The three main criteria of CAD are reduction in the minimal erythema dose to UVA, UVB and/or visible light; a persistent eczematous eruption that predominantly affects sun-exposed skin and sometimes extends to covered areas; and histopathologic changes consistent with chronic eczema with or without cutaneous lymphoma-like changes [6].
Chronic actinic dermatitis has been reported in Caucasians, Latin Americans, Blacks, Japanese, and Indians and is most common in temperate climates [7, 8]. The condition is typically seen in elderly men and is rare in women [7]. It may or occur after endogenous eczema, photoallergic or allergic contact dermatitis, oral drug photosensitivity, occasionally polymorphic light eruption, or rarely human immunodeficiency virus infection [8].
The initial symptom is usually persistent erythema of the face, with subsequent development of an eczematous, patchy or diffuse, pruritic, often lichenified eruption in sun-exposed areas, particularly the face, scalp, V-area of chest, back and sides of neck, and dorsal aspects of hands and forearms, often with sharp demarcation at lines of clothing [8]. After some time, and often rapidly, non-sun-exposed areas also become involved [7]. Palmar and plantar eczema is also possible as well as loss of eyebrow and scalp hair that develops after scratching; erythroderma occasionally may develop [8]. Affected individuals with CAD may not complain of abnormal reactions to sunlight because they may be so sensitive to UVA and visible wavelengths, which penetrate window glass, that no relationship to sunlight exposure is noticed [9].
The histopathologic changes are characterized by epidermal spongiosis and acanthosis, sometimes with hyperplasia, along usually with a deep dermal, predominantly perivascular, frequently dense, mononuclear-cell infiltrate and not infrequently large hyperchromatic convoluted nuclei and mitotic figures. There also may be macrophages, eosinophils, and plasma cells; in some instances, the disorder may be difficult to differentiate from cutaneous T-cell lymphoma (CTCL) [8]. Chronic actinic dermatitis has a characteristic immunophenotype distinct from most cases of CTCL, with CD8 cells in the epidermis and discordance between BF1 (ß-chain constant region of T-cell receptor) cells and CD3 expression in CAD [10].
Phototests to ultraviolet and often visible irradiation may show erythematous or eczematous responses, normally at doses much lower than the normal minimal erythema dose. The reaction peaks between seven and 24 hours after exposure. The provoking wavelengths are ultraviolet B (UVB) (280-320 nm) in virtually all patients, ultraviolet A (UVA) (320-400 nm) in most patients, and visible light or UVA alone in a few patients [9, 8].
Allergic and/or photallergic contact dermatitis commonly coexist with CAD and often precedes the onset of photosensitivity [6]. In 1 study of 89 patients with CAD, 74 percent had positive patch or photopatch tests. Of patch tests 36 percent were positive to sesquiterpene lactone mix, 21 percent to fragrance compounds, 20 percent to colophony, and 14 percent to rubber chemicals. In 1 photopatch test study, 6 percent were positive to musk ambrette, 7 percent to sunscreens, and 1 to both [11].
Other tests to consider are ANA and anti-Ro and anti-La antibodies [8].
The pathogenisis of CAD is not completely understood, but the histologic and immunohistochemical features of CAD, along with increased ICAM-1 expression and a dermal infiltrate of predominantly CB8+ T cells mimic the delayed-type hypersensitivity response observed in contact dermatitis [8, 6]. One theory has proposed that during an initial photoallergic reaction, a normal skin constituent is altered to become antigenic. Induction begins with UVA-dependent covalent binding of hapten to an endogenous protein and is followed by a delayed-type hypersensitivity response. As the disease progresses to CAD, UVB and UVA alone may trigger the immune response without the hapten by continuing to form the antigenic photoproduct from the endogenous carrier protein [6]. There has been theoretical support for this mechanism using an in vitro model with the photosensitizer tetrachlorosalicylanilide, which demonstrated phototoxic oxidation of histidine with modification of the carrier albumin into a weak antigen [12].
The mainstay of treatment is sunlight avoidance and, when applicable, allergen avoidance. Sunlight avoidance includes wearing protective clothing, application of non-irritating broad-spectrum sunscreens, and application of filters to home and car windows. Topical and/or oral glucocorticoids with emollients are usually necessary [8]. Topical tacrolimus ointment has been shown to be beneficial in case reports [13, 14]. Treatments for resistant disease include low-dose PUVA photochemotherapy, narrow-band UVB phototherapy, cyclosporine, azathioprine, and mycophenolate mofetil alone or in combination [8, 15].
Resolution of photosensitivity in 1 study of CAD occurred in 20 percent in 10 years; severe abnormal UVB photosensitivity and positive patch tests to 2 or more unrelated patch test batteries to be predictors of a poorer prognosis [16]. There is usually persistence of the allergic contact allergy even with clinical improvement in the photosensitivity [17].
References
1. Hawk JLM, Magnus IA. Chronic actinic dermatitis-an idiopathic photosensitivity syndrome including actinic reticuloid and photosensitive eczema. Br J Dermatol 1979;101(Suppl 17):242. Wilkinson DS. Photodermatitis due to tetrachlorosalicylanilide. Br J Dermatol 1961;73:213
3. Ive FA, et al. 'Actinic Reticuloid'; a chronic dermatosis associated with severe photosensitivity and the histological resemblance to lymphoma. Br J Dermatol 1969;81:469
4. Ramsay CA, Black A. Photosensitive eczema. Trans St. John's Hosp Derm Soc 1973;59:152
5. Fain-Bell W, et al. The syndrome of chronic photosensitivity dermatitis and actinic reticuloid. Br J Dermatol 1974;91:617
6. Somani VK. Chronic actinic dermatitis-a study of clinical features. Indian J Dermatol Venereol Leprol 2005;71:409
7. Roelandts R. Chronic actinic dermatitis. J Am Acad Dermatol 1993;28:240
8. Hawk JLM. Chronic actinic dermatitis. Photodermatol Photoimmunol Photomed 2004;20:312
9. Dawe R. Chronic actinic dermatitis in the elderly. Drugs Aging 2005;22:201
10. Heller P, et al. Chronic actinic dermatitis: an immunochemical study of its T-cell antigenic profile, with comparison to cutaneous T-cell lymphoma. Am J Dermatopathol 1994;16:510
11. Menage H, et al. Contact and photocontact sensitization in chronic actinic dermatitis: sesquiterpene lactone mix is an important allergen. Br J Dermatol 1995;132:543
12. Kochevar IE, Harber LC. Photoreactions of 3,3',4',5-tetrachlorosalicylanilide with proteins. J Invest Dermatol 1977;68:151
13. Schuster C, et al. Successful treatment of recalcitrant chronic actinic dermatitis with tacrolimus. Dermatology 2004;209:325
14. Baldo A, et al. A case of chronic actinic dermatitis treated with topical tacrolimus. J Derm Treat 2005;16:245
15. Nousari HC, et al. Mycophenolate in psoralen-UV-A desensitization therapy for chronic actinic dermatitis. Arch Dermatol 1999;135:1128
16. Dawe RS, et al. The natural history of chronic actinic dermatitis. Arch Dermatol 2000;136:1215
17. Addo HA, Frain-Bell W. Persistence of allergic contact sensitivity in subjects with photosensitivity dermatitis and actinic reticuloid syndrome. B J Dermatol 1987;117:555
© 2008 Dermatology Online Journal

