Paradoxical effects of cost reduction measures in managed care systems for treatment of severe psoriasis
Published Web Location
https://doi.org/10.5070/D30wz4h31bMain Content
Paradoxical effects of cost reduction measures in managed care systems for treatment of severe psoriasis
Brad A Yentzer MD1, Christopher B Yelverton MD MBA1, Gregory L Simpson1, Jason F Simpson MSA CPA1, Wenke Hwang PhD3, Rajesh Balkrishnan PhD4, Steven R Feldman MD PhD1, 2, 3
Dermatology Online Journal 15 (4): 1
1. Center for Dermatology Research, Department of Dermatology2. Center for Dermatology Research, Department of Pathology
3. Center for Dermatology Research, Department of Public Health Sciences
Wake Forest University School of Medicine, Winston-Salem, North Carolina. sfeldman@wfubmc.edu
4. Ohio State University College of Pharmacy and School of Public Health, Columbus, Ohio
Abstract
Background: Insurance companies vary widely in their coverage policies for severe psoriasis therapies. Unfortunately, coverage policies for psoriasis therapies do not necessarily follow current treatment paradigms, such that more expensive second or third line treatments may be more easily obtained than first line treatments. Methods: We reviewed insurance policy bulletins, statements of coverage/medical necessity, and prior authorization forms for three large insurance carriers regarding psoriasis treatment with biologic agents and phototherapy. A cost comparison was performed to estimate total costs to patients and insurer under the current system as well as a hypothetical system in which co-pays and deductibles are eliminated. Additionally, we reviewed the total cost to an insurer for placing a patient on a trial of home phototherapy before approving use of expensive biologics. Results: Requirements for coverage for phototherapy treatments are often the same, if not more stringent, than those for biologics. On an annual per patient basis, insurance companies pay an estimated $5, $76, and $23,408 for home phototherapy, office phototherapy, and biologics, respectively. The first year cost to patients, however, is estimated to be $2,590, $3,040, and $920 for home phototherapy, office phototherapy, and biologics, respectively. An initial 3-month trial of home phototherapy yields a graded annual cost savings to insurers of $21,610 to $2,110 per patient. Discussion: The evolution of psoriasis treatment has resulted in a paradoxical situation in which the use of lower-cost psoriasis treatments, with longer safety track records, is discouraged relative to newer options. If co-pays, deductibles, and prior authorization requirements that discourage phototherapy were reduced or eliminated, more patients and physicians would likely choose phototherapy over biologics. This has the potential to reduce overall healthcare costs for psoriasis management.
Introduction
Psoriasis is a complex, lifelong disease that affects over 5 million Americans. Mild and localized psoriasis is often treated with topical agents (steroids and vitamin D derivatives). However, for the approximately 1.5 million people with more severe or extensive forms of disease, therapy often involves a combination of topical, phototherapy, and systemic therapies (such as methotrexate, acitretin, and injected/infused immunomodulators or "biologics"). Although there are multiple treatment options and several different strategies for implementing them, the goals of treatment are: to gain control of the disease quickly and effectively; to decrease scaling, erythema, and the thickness of the individual plaques; to decrease total body surface area involvement; to maintain remission and avoid relapse; to minimize side effects; and most importantly, to improve a patient's quality of life.
In published treatment algorithms, phototherapy is a first choice for patients with moderate to severe psoriasis in which application of topical medications is impractical [1, 2, 3]. Ultraviolet phototherapy is one of the safest and most effective therapies for severe psoriasis [3]. Broad-band UVB, narrow-band UVB, or PUVA can be used. Whereas most phototherapy is delivered in a physician's office, home UVB devices are also available. Home phototherapy is more convenient and less costly compared to office-based phototherapy or other systemic treatments [4].
Insurance companies vary widely in their coverage policies for severe psoriasis therapies. The range of treatment options may fall under multiple portions of a patient's health benefits package (medical, pharmacy, durable medical equipment), with some options not being covered at all. These variations in coverage options may be a significant hindrance to the selection of an ideal treatment plan. Strangely enough, more costly treatments with less long-term safety data, such as biologics, may be more easily obtained under some plans than other well-studied, less-expensive options, such as phototherapy. To better understand these paradoxical effects, we explore and summarize prior authorization requirements of three insurers for treating severe psoriasis with phototherapy and biologics.
Methods
We reviewed insurance policy bulletins, statements of coverage/medical necessity, and prior authorization forms for three large insurance carriers (Aetna, Cigna, and BCBS of California) regarding psoriasis treatment with biologic agents and phototherapy. These polices are available online via the National Psoriasis Foundation [5]. As part of an effort to illustrate the difficulties many patients have obtaining coverage for appropriate treatments, specific attention was placed on insurers' requirements for coverage of each therapy [6-11].
Since patients are influenced not only by the availability of coverage, but by the out of pocket expenses of treatments, we analyzed the cost to both patients and insurance companies for office phototherapy, home phototherapy, and biologics (using etanercept as our example). To calculate this, we made a few assumptions: the annual number of office visits required is 2 when using home phototherapy, 76 when using office phototherapy (based on 3 visits per week x 12 weeks to achieve a remission and once per week x 40 more weeks for maintenance) [12], and 8 when using etanercept; the co-pay for each office visit was based on Aetna's California Open Access plans [3]; the office cost billed to insurers was based on the 2007 Medicare fee schedule for a level 3 return (R3) visit [3, 5]; the co-pay for etanercept is $50 per month; and the deductible for a home phototherapy unit is $2,000. The total out-of-pocket expense seen by the patient is the sum of co-pays and deductibles for one year. The calculated cost to insurers for each treatment equals the sum of office and medication costs, minus co-payments made by the patient.
We then used flow charts to demonstrate how co-payments and deductibles might influence patient behavior and, ultimately, the total cost to insurance companies for covering 1,000 psoriasis patients. Up to 20 percent of psoriasis patients may have psoriatic arthritis, some of whom may require systemic therapy [1]. Although it is likely an over-estimation, for our purposes we will assume that all 20 percent will be placed on biologics.
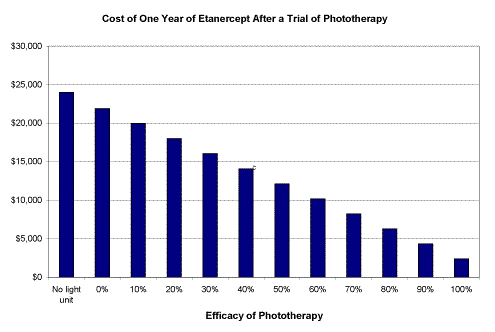
Lastly, we analyzed the cost benefit of a 3-month trial of home phototherapy before initiating a biologic. Since phototherapy is not effective for all psoriasis patients, we calculated the savings to insurers based on the efficacy of the phototherapy trial and presume that greater efficacy equals longer time before initiating a biologic. For this calculation, we used a few assumptions: Etanercept costs $3,000 for double dosing the first 3 months and $1,500 per month for single dosing thereafter [14]; a 6 foot (6 bulb, single panel) home phototherapy unit costs a $2,390 one time expense [15]; the efficacy of a light unit = p; and the cost for managing a patient for one year will = cost of the light unit + (1-p) x cost of Etanercept. Using this calculation, if the phototherapy is not effective at all, the patient will begin therapy at month 4; whereas if it is 100 percent effective, there is no need for a biologic.
Results
Specific requirements for treatment coverage vary between carriers. Both Aetna and BCBS of California have no specific requirements stated in their policies for coverage of office based UVB phototherapy. All 3 major carriers appear to have fewer requirements for office phototherapy than biologics. Both BCBS and Cigna also appear to have fewer requirements for biologics than home phototherapy, making home phototherapy the most difficult to get approved (Table 1).
|
|
| Figure 1 | Figure 2 |
|---|---|
|
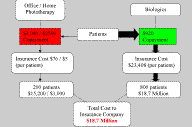
Figure 1. Patient Behaviors as a Result of Copayments for Office-Based Phototherapy. The high copayments from multiple office visits and high out-of-pocket deductibles deter many patients from using phototherapy,
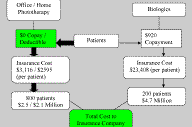
and guide them toward the use of expensive biologics. Figure 2. Patient Behaviors in the Absence of Copays or Deductibles for Office or Home Phototherapy. For Office Phototherapy, total annual copay is estimated to be $3,116 (see Table 2). For Home Phototherapy, $2,590 equals the cost of average light unit + estimated annual copay ($2,390 + $200) [15, 17]. Assume 20% of patients will have psoriatic arthritis or otherwise need systemic treatment with biologics and that the insurance company would cover 100% of the cost of a home light unit. With less out-of-pocket expense, more patients would likely choose phototherapy over treatment with biologics, and potentially save the health care system over $10 million annually (compare to Fig. 1). |
|
When comparing treatments on an annual per patient basis (Table 2), insurance companies pay an estimated $5, $76, and $23,408 for home phototherapy, office phototherapy and biologics, respectively. The first year cost to patients is estimated to be $2,590 ($200 co-pay + $2,390 deductible for light unit), $3,040, and $920 for home phototherapy, office phototherapy and biologics, respectively. Assuming that at least 80 percent of patients given this paradigm would select biologics, an annual estimated cost to insurers to cover 1,000 patients would be over $18 million (Fig. 1). If insurers covered 100 percent of the co-pays and deductibles for phototherapy, we would assume that only 20 percent (those with psoriatic arthritis) would remain on biologics and cost insurers an estimated $6 to $7 million per year per 1,000 patients (Fig. 2). This change in policy has the potential to save insurers over $10 million annually.
|
| Figure 3 |
|---|
| Assumptions: Etanercept costs $3,000 for double dosing the first 3 months and $1,500 per month for single dosing thereafter [14]; a 6 foot home phototherapy unit costs a $2,390 one time expense; the efficacy of a light unit = p; and cost for managing a patient for one year will = cost of light unit + (1-p) x cost of etanercept. Using this calculation, a 3-month trial of home phototherapy is less expensive than starting immediately with biologics, even if the phototherapy is 0% effective. Assuming an 80% success rate for one year with home light, companies can potentially save over $17,000 per patient for the first year of treatment by applying a 3-month phototherapy trial to the treatment paradigm [18]. |
Analysis of the cost to insurers for placing all psoriasis patients on a home phototherapy unit for at least 3 months before attempting biologics shows a graded annual cost savings of $21,610 per patient, if the phototherapy is 100 percent effective, to $2,110 per patient, if the phototherapy is not effective at all (Fig. 3). This means that insurers would profit from facilitating patients to try home phototherapy for 3 months before biologics, regardless of a patient's response (or adherence) to phototherapy.
Discussion
Many policies regarding prerequisites for psoriasis treatments are vague and it should be noted that clarification of these policies from the insurers was not easily accessible over the phone. Requirements for treatment with home-based phototherapies appear to be more stringent under some plans than the requirements for newer biologic (injected/infused immunomodulator) treatment options. Such policies may hinder access to appropriate treatment and shift patients toward more expensive treatments with less long-term safety and efficacy data. The coverage policies that discourage the use of phototherapy are exacerbated by out-of-pocket/co-payment disincentives that further deter patients' use of phototherapy (Fig. 1).
If insurance companies provided incentives for home phototherapy, it would benefit both patients and the insurers by reducing overall costs to both insurers and patients (Fig. 2). Such incentives could include fully covering the cost of a home light unit or eliminating office visit co-pays for office-based phototherapy. Additionally, policies need to be made crystal clear about exactly what they cover. Step-by-step instructions on how to get coverage or reimbursement for the desired treatment should be provided.
Our understanding of the severity of psoriasis and its impact on patients' lives has evolved along with the development of new treatment options. A clear standard has developed such that greater than 10 percent affected body surface area is sufficient (but not necessary) for declaring patients to have severe disease. Clinical trials of new biologics for psoriasis have used this as one criterion for eligibility. Policies for coverage of biologics have also adopted this standard. However, policies for home-based phototherapy coverage suffer from outmoded definitions of severity, equating severe with greater than 30 percent body surface area. Along with the frequently required "proof of efficacy of phototherapy" in a physician's office, these additional requirements have paradoxically made it harder for patients to access phototherapy compared to accessing the new biologic treatments. Requirements for the demonstration of efficacy of office-based phototherapy prior to prescription of a home phototherapy unit is counter productive. Given that a single month's supply of a biologic may exceed the cost of a home phototherapy unit, this restriction is not rational. However, some physicians may feel uncomfortable with immediately initiating home phototherapy in certain patients without first assessing how that particular patient will comply with and respond to phototherapy. In these cases, it is reasonable to initiate office phototherapy prior to home UV phototherapy.
The insurance plans examined do not represent an exhaustive list and coverage may vary among different plans offered by a carrier. Additionally, we did not examine a physician's probability to prescribe one treatment modality over another. However, we can assume that insurance prerequisites also influence providers, such that the physicians are less likely to prescribe a treatment such as home phototherapy if they feel it is a hassle to get. Even if one assumes that patients would opt for home phototherapy if it did not require prerequisites for coverage or costly deductibles, phototherapy is not effective for all patients. However, as demonstrated, a 3-month trial of home phototherapy before treating with biologics reduces overall costs and is more in line with current treatment paradigms.
References
1. Weinstein GD, Gottlieb AB, National Psoriasis Foundation (U.S.). Therapy of moderate-to-severe psoriasis. New York: ; M. Dekker, 2003.2. Feldman S.R., Koo J.Y., Lebwohl M.G., Menter A., Van Voorhees A. The Psoriasis and Psoriatic Arthritis Pocket Guide: Treatment Algorithms and Management Options. 2005. National Psoriasis Foundation. Ref Type: Pamphlet
3. Feldman SR, Garton R, Averett W, Balkrishnan R, Vallee J. Strategy to manage the treatment of severe psoriasis: considerations of efficacy, safety and cost. Expert Opin Pharmacother 2003;4(9):1525-33.
4. Yelverton CB, Kulkarni AS, Balkrishnan R, Feldman SR. Home ultraviolet B phototherapy: a cost-effective option for severe psoriasis. Manag Care Interface 2006;19(1):33-6, 39.
5. National Psoriasis Foundation. Online Major Health Plans' Insurance Policies. http://www.psoriasis.org/medical/practice/insurers/links.php. Accessed: 8-17-2007.
6. Aetna. Clinical Policy Bulletin: Phototherapy and Photochemotherapy (PUVA) for Skin Conditions. http://www.aetna.com/cpb/medical/data/200_299/0205.html. Accessed: 7-27-2007.
7. Aetna. Clinical Policy Bulletin: Psoriasis: Biological Therapies. http://www.aetna.com/cpb/medical/data/600_699/0658.html. Accessed: 7-27-2007.
8. Blue Cross of California. Clinical UM Guideline: Biologics for Psoriasis and Psoriatic Arthritis. http://medpolicy.bluecrossca.com/policies/ guidelines/DRUG/psoriasis_arthritis.html. Accessed: 7-27-2007.
9. Blue Cross of California. Medical Policy: Ultraviolet Light, Including Laser Therapy, for the Treatment of Skin Disorders (MED.00008). http://medpolicy.bluecrossca.com/policies/ MED/UV_skin_disorders.html. Accessed: 7-27-2007.
10. CIGNA. Healthcare Coverage Position: Phototherapy and Photochemotherapy for Dermatological Conditions. http://www.cigna.com/customer_care/healthcare_professional/ coverage_positions/medical/ mm_0031_coveragepositioncriteria_phototherapy_ and_photochemotherapy.pdf. Accessed: 7-27-2007.
11. CIGNA. Healthcare Coverage Position: Etanercept. http://www.cigna.com/customer_care/healthcare_professional/ coverage_positions/pharmacy/ ph_5111_coveragepositioncriteria_etanercept_enbrel.pdf. Accessed: 7-27-2007.
12. Stern RS, Armstrong RB, Anderson TF, Bickers DR, Lowe NJ, Harber L et al. Effect of continued ultraviolet B phototherapy on the duration of remission of psoriasis: a randomized study. J Am Acad Dermatol 1986;15(3):546-52. [PubMed]
13. Aetna. Plans at a Glance in California. http://www.aetna.com/members/individuals/health/plans.html. Accessed: 8-10-2007.
14. Epocrates Online. The Cost of Etanercept. https://online.epocrates.com. Accessed: 7-27-2007.
15. HomePhotoTherapy.com. Panosol II - Six Foot - Six Lamp Pricing. http://www.homephototherapy.com/UV-PRICES.htm. Accessed: 6-10-2008 .
16. Aetna. Plans at a Glance in North Carolina. http://www.aetna.com/members/individuals/health/ plan_details/NorthCarolina/. Accessed: 8-10-2007.
17. Sander HM, Morris LF, Phillips CM, Harrison PE, Menter A. The annual cost of psoriasis. J Am Acad Dermatol 1993;28(3):422-5. [PubMed]
18. Camisa C. Handbook of psoriasis. Malden, Mass.: ; Blackwell Pub., 2004.
© 2009 Dermatology Online Journal

