Cutaneous mastocytosis: Two pediatric cases treated with topical pimecrolimus
Published Web Location
https://doi.org/10.5070/D30fk211zhMain Content
Cutaneous mastocytosis: Two pediatric cases treated with topical pimecrolimus
Osvaldo Correia MD PhD1,2, Ana Filipa Duarte MD1, Paula Quirino MD1, Rosa Azevedo MD3, Luis Delgado MD PhD2
Dermatology Online Journal 16 (5): 8
1. Centro de Dermatologia Epidermis, Instituto CUF, Porto, Portugal. epidermis@epidermis.pt2. Department of Immunology, Faculty of Medicine, University of Porto, Portugal
3. Department of Pathology, Institute of Cancer, Porto, Portugal
Abstract
Cutaneous mastocytosis is characterized by increased numbers of skin mast cells that release mediators causing pruritus, urticaria, and flushing. Most pediatric mastocytosis patients exhibit the pattern of urticaria pigmentosa, which typically appears during the first two years of life and resolves spontaneously in late adolescence. However, while the disease is active, patients are frequently symptomatic and uncomfortable, which justifies symptomatic treatment. We report 2 patients, a 14-month-old girl and a 26-month-old boy, with localized cutaneous erythematous lesions with a positive Darier sign. In each, a punch biopsy confirmed the diagnosis of mastocytosis. Treatment was instituted with pimecrolimus cream twice a day and oral antihistamine. An almost complete response was achieved after 4 months of therapy in both patients, with no clinical evidence of recurrence after 4 years and 2 years of follow-up, respectively. In children, the treatment of mastocytosis is directed primarily to avoiding potential mast cell degranulating agents and alleviating symptoms. Topical calcineurin inhibitors act by inhibiting T-cell activation and cytokine release; they may suppress mast cell- mediated reactions by reducing their degranulation. These two cases suggest that in localized cutaneous mastocytosis they are a safe and efficacious alternative to topical steroid therapy.
Introduction
Mastocytosis represents a spectrum of rare clinical disorders characterized by an increase in tissue mast cells [1, 2]. It is traditionally divided into cutaneous mastocytosis and systemic mastocytosis. Cutaneous mastocytosis can be divided into four clinical variants: urticaria pigmentosa, solitary mastocytoma, diffuse cutaneous mastocytosis, and telangiectasia macularis eruptive perstans [3].
The onset of mastocytosis can occur from birth to adulthood, but childhood disease is more common and often affects only the skin. Most pediatric mastocytosis cases exhibit the form of urticaria pigmentosa that typically appears during the first two years of life and resolves spontaneously in late adolescence [1, 2].
Mutations of the c-kit proto-oncogen, coding for the transmembrane receptor kit of the stem cell growth factor, have been implicated in the etiology of mastocytosis. The stem cell growth factor has a role in maturation, proliferation, and activation of mast cells, which produce tryptase [4]. Total serum tryptase correlates with the extent of the mast cell disease [5].
Case report
We report 2 patients with cutaneous mastocytosis.
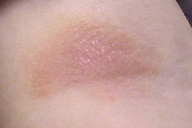
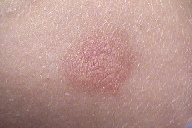
Patient 1 is a 14-month-old girl with a limited cutaneous erythematous plaque located on the right upper arm, which showed a positive Darier sign (urtication induced by stroking lesions) (Figure 1a). The plaque appeared in the first 4 months of life.
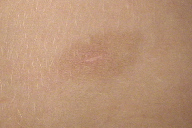
Patient 2 is a 26-month-old boy with erythematous papules located on the right lower back that evolved over several months (Figure 2a).
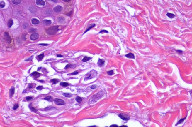
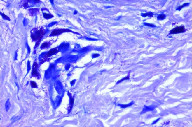
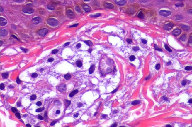
The personal and family histories of both patients were non-contributory. Systemic symptoms, including abdominal pain, diarrhea, palpitations, syncope, bone pain, or weigh loss were absent in both children. Total serum tryptase was normal in both and the clinical diagnosis was mastocytosis in both cases. A punch biopsy was performed in each patient that revealed telangiectasia and perivascular mast cells in H&E. Giemsa staining confirmed the presence of granules (Figures 1b, 1c, 2b, and 2c).
 |  |
| Figure 2c | Figure 2d |
|---|---|
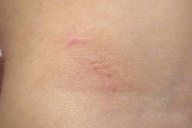
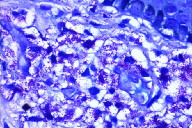
| Figure 2c. Patient 2: The Giemsa stain highlights the cytoplasmic granules typical of mast cells (x40). Figure 2d. Patient 2: Complete resolution after 6 months of treatment. | |
Treatment was instituted with pimecrolimus cream (twice a day during three months, followed by once a day until at least 2 months after clinical resolution) and oral antihistamine (loratadine, 5 mg once a day). Almost complete resolution was achieved after 4 months of therapy in both patients (Fig 1d and 2d). Patient 1 used pimecrolimus for a total of 9 months and patient 2 for a total of 6 months. During follow-up there was no clinical evidence of recurrence after 4 years in patient 1 and 2 years in patient 2.
Discussion
The prognosis of pediatric mastocytosis is good; the children are usually without systemic involvement and complete spontaneous resolution is common in months to years [1, 3]. However, while the disease is active, patients are frequently symptomatic and uncomfortable, which justifies symptomatic treatment [4]. The occurrence of pruritus, urtication and flushing caused by mediator release can be reduced by avoiding potential mast cell degranulating agents, such as certain food and drugs, exercise, heat, and friction.
Because histamine is the single most important mediator involved, antihistamines are commonly used as first-line treatment to alleviate symptoms. Topical steroids have been proposed but the outcome is variable [6].
The topical calcineurin inhibitors (TCI) have substantially improved the treatment of atopic dermatitis and also have a beneficial effect in other inflammatory dermatoses such as seborrheic dermatitis, genital lichen sclerosus, intertriginous psoriasis, cutaneous lupus erythematosus, autoimmune bullous dermatosis, vitiligo, graft-versus-host disease, and Netherton syndrome [7].
Topical calcineurin inhibitors (TCI) act by inhibiting T-cell activation and cytokine release, but they also reduce pruritus and erythema, which canÕt be explained by T-cell interactions alone. On the other hand, there is evidence they transiently induce neuropeptide release, namely substance P and calcitonin gene-related peptide, that bind to mast cells leading to their degranulation. This explains the abnormal transient burning, stinging, and itching at the beginning of therapy with TCI [8]. However, in an experimental model, the continuous application of topical tacrolimus suppresses mast cell-mediated reactions by reducing their degranulation rather than by decreasing their number [9].
Although pediatric cutaneous mastocytosis may resolve spontaneously, the quick and sustained response of these two cases corroborates the effectiveness of the use of TCI in the treatment of mast cell-dependent diseases such as mastocytosis. Furthermore, we have also used TCI for the treatment of four more children with urticaria pigmentosa, with the same regimen (pimecrolimus cream twice a day during three months followed by application once a day, until 2 months after clinical resolution and oral antihistamine - loratadine, 5 mg once a day). In these four patients (all males, ages ranging from 7 to 16 months), all with normal serum tryptase, significant improvement was achieved 3 months after starting treatment (unpublished data).
There are few reports of cutaneous mastocytosis treated with TCI [10, 11]. Concerning their advantages in relation to topical steroids, we emphasize the more selective mechanism of action, the absence of skin atrophy, and the lack of significant systemic absorption [7]. With this profile they could be a low toxicity first line therapy for the symptoms of cutaneous mastocytosis,and an alternative to topical corticosteroids. However, because the majority of children who develop urticaria pigmentosa before the age of two years resolve spontaneously, large-scale randomized placebo-controlled studies are needed to clarify the role of TCI in the treatment of cutaneous mastocytosis.
References
1. Mastocytosis in children: a protocol for management. Heide R, Beishuizen A, De Groot H, Den Hollander JC, Van Doormaal JJ, De Monchy JG, Pasmans SG, Van Gysel D, Oranje AP; Dutch National Mastocytosis Work Group. Pediatr Dermatol. 2008:25(4):493-500. [PubMed]2. Akoglu G, Erkin G, Cakir B, Boztepe G, Sahin S, Karaduman A, Atakan N, Akan T, Kolemen F. Cutaneous mastocytosis: demographic aspects and clinical features of 55 patients. J Eur Acad Dermatol Venereol 2006;20:969-973. [PubMed]
3. Kiszewski AE, Durán-Mckinster C, Orozco-Covarrubias L, Gutiérrez-Castrellón P, Ruiz-Maldonado R. Cutaneous mastocytosis in children: a clinical analysis of 71 cases. J Eur Acad Dermatol Venereol 2004;18:285-290. [PubMed]
4. Flageul B. Cutaneous mastocytosis. Rev Prat 2006;31:1745-1751. [PubMed]
5. Valent P, Escribano L, Parwaresch R, Schemmel V, Schwartz LB, Sotlar K, Sperr WR, Horny HP. Recent advances in mastocytosis research. Summary of the Vienna Mastocytosis Meeting 1998. Int Arch Allergy Immunol 1999;120:1-7. [PubMed]
6. Heide R, de Waard-van der Spek FB, den Hollander JC, Tank B, Oranje AP. Efficacy of 25% dilute fluticasone propionate 0.05% cream as wet-wrap treatment in cutaneous mastocytosis. Dermatology 2007;214:333-335. [PubMed]
7. Luger T, Paul C. Potential new indications of topical calcineurin inhibitors. Dermatology 2007;215 suppl 1:45-54. [PubMed]
8. Ständer S, Ständer H, Seeliger S, Luger TA, Steinhoff M. Topical pimecrolimus and tacrolimus transiently induce neuropeptide release and mast cell degranulation in murine skin. Br J Dermatol 2007;156:1020-1026. [PubMed]
9. Inoue T, Katoh N, Kishimoto S. Prolonged topical application of tacrolimus inhibits immediate hypersensitivity reactions by reducing degranulation of mast cells. Acta Derm Venereol 2006;86:13-16. [PubMed]
10. Avshalumov K, Pichardo R, Jorizzo JL, Sangueza OP, Goldenberg G. Bullous mastocytosis: report of a patient and a brief review of the literature. Am J Dermatopathol. 2008 Oct;30(5):455-7. [PubMed]
11. Yoko N, Yasuyuki S, Hiroyuki M, Chika O, Ichiro K, Tatsuhiko F. Successful Treatment with FK506 Ointment for Adult-onset Mastocytosis; A Case Report. Jap J Dermatol 2005;115:2232-2235
© 2010 Dermatology Online Journal