Management of skin ulcers in a patient with mycosis fungoides
Published Web Location
https://doi.org/10.5070/D30882t9vxMain Content
Management of skin ulcers in a patient with mycosis fungoides.
Vincenzo Panasiti1, Valeria Devirgiliis1, Riccardo G Borroni1, Mariarita Rossi1, Michela Curzio1, Monica Mancini1, Ugo Bottoni2, and Stefano Calvieri1
Dermatology Online Journal 12 (2): 16
1. Department of Dermatology, University "La Sapienza" - Rome, Italy. ilcapo75@gmx.net2. Department of Dermatology, University "Magna Graecia" - Catanzaro, Italy
Abstract
We present a patient with a cutaneous T-cell lymphoma/mycosis fungoides (CTCL/MF) followed for more than 10 years. After several different aggressive treatments to control progression of CTCL/MF, the patient developed several ulcerated tumors on the abdomen and limbs. Specific systemic antibiotic therapy failed to treat skin infection. While treating the stage III CTCL with polychemotherapy, we used an active colloidal hydrogel topically to manage wound healing and to treat and prevent potential sources of sepsis. After 11 weeks of treatment we observed complete cicatrization of ulcerated tumors. We reported on this case to describe the importance of a correct management of skin ulcers in immunosuppressed patients in order to avoid possible systemic spread of infection which represents the major cause of death in these patients.
Cutaneous T-cell lymphoma (CTCL) is the most common primary lymphoma of the skin. As with most cancers, survival depends on the stage of disease. Infection is a common complication of CTCL. Infection continues to be a major cause of both morbidity and mortality in the cancer patient because of alterations either in the host's overall health or in the individual's state of immunity [1, 2, 3]. We report on a patient with advanced mycosis fungoides (MF) complicated by infected ulcerated lesions.
Clinical synopsis
A 43-year-old man presented to our department of dermatology in November 1991 for multiple erythematous oval patches and plaques that appeared on his trunk, arms, and legs 6 months earlier. According to the clinical picture and the histopathological features, a diagnosis of cutaneous T-cell lymphoma (CTCL)/mycosis fungoides (MF), plaque stage was made. Treatment with lymphoblastoid interferon-alpha (IFN-α) at the dosage of 1,000,000 IU three times a week was started, achieving partial response after 3 months. From February 1992 to March 1997 the patient came to control irregularly, generally after evident relapses of the disease. However, this treatment had to be repeatedly stopped for the constant worsening of diabetes he had been suffering from for more than 10 years.
In April 1997 nodules appeared on his lower limbs. Histopathology of a skin biopsy specimen revealed tumor-stage MF. Therapy was started with IFN-α 2a and etretinate 0.5-1 mg/kg/ per day given orally, achieving partial response with the complete regression of nodular lesions. The treatment was continued for 1 year. In December 1998 the patient presented with a new relapse. Four courses of oral 8-methoxypsoralen plus UVA irradiation (PUVA) were administered, followed by IFN-α 2a and etretinate, achieving partial response. From September 1999 to December 1999 he received two courses of extracorporeal photopheresis to treat an aggressive relapse obtaining minor response. In September 2001 the patient presented again with nodular lesions with ulcerations localized on his trunk and limbs. He also had bilateral inguinal lymphadenopathy, measuring up to 2.5 cm. One lymph node was excised surgically from the inguinal region, and histopathologic diagnosis was dermatopathic lymphadenopathy. A total-body CT scan and a bone marrow biopsy were performed for staging purposes and, as in the past, they were negative for localizations of lymphoma. A total-skin electron beam irradiation (TSEBI) was performed achieving an evident clinical partial response, with complete regression of the nodular component. In November 2003, MF progressed again presenting several widespread skin tumors on trunk and limbs. Some of the tumors were ulcerated, including a large tumor on the abdomen producing a bad smelling discharge (Fig. 1). The patient was hospitalized and a swab was positive for coagulase-negative Staphylococcus; but the patient failed to respond to specific oral antibiotic treatment. Meanwhile, a polychemotherapy regimen was started with liposomal doxorubicin 20 mg/m², bleomicin 10 mg, vinblastin 10 mg, dacarbazine 600 mg, betamethasone 6 mg all intravenously per day every 15 days (ABVD modified). Prednisone 25 mg daily per os was also administered in the intervals. During this period we used an active colloidal hydrogel topically (Flamigel®, Flen Pharma, Antwerp-Belgium) for the management of the ulcerative lesions to promote skin regeneration and to avoid infections. After accurate washing with saline solution, wounds were gently dried with a sterile gauze, then active colloidal hydrogel was applied in a 5 mm-thick layer and covered with vaseline gauze. Medication was repeated every 4 days. Complete cicatrization of skin ulcer on the abdomen was achieved after 11 weeks of treatment (Fig. 2). After 13 courses of polychemotherapy the nodules on the left leg, trunk and abdomen completely regressed.
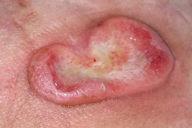 | 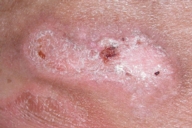 |
| Figure 1 | Figure 2 |
|---|
Discussion
CTCL is a hematologic malignancy that may result in alteration of the immune system, especially the skin immune system (SIS). Hypothetically the alteration of SIS may potentiate local infectious processes. The infectious complications of CTCL are of paramount importance because they are involved in over 50 percent of deaths in patients with CTCL [4, 5, 6]. Cutaneous infections are the most frequent infectious complications of CTCL, and, although rarely fatal, they represent the most common source of sepsis [6, 7]. Alteration of SIS may also imply a systemic immunosuppression; abnormalities of lymphocyte subsets in peripheral blood of patients with advanced-stage CTCL are well documented [8, 9]. Therefore the reasons for the increased susceptibility to infection by patients with CTCL is partially the result of disruption of the normal skin barrier. Disruptions in the integument occur with tumor infiltration and are often accompanied by ulceration, necrosis, and invasion of pathogenic bacteria [6]. In a retrospective study of 356 CTCL patients, 478 infections were documented, with the skin being the most commonly infected site by far, followed by blood and lungs [6]. In addition to being most frequently infected, the integument is reported as the source of sepsis in up to 80 percent of cases [7]. Bacteremia and bacterial pneumonias have been described as the most common infectious causes of death in CTCL patients [6, 10].
In our patient the treatment of MF with polychemotherapy induced remission of ulcerated tumors. Furthermore, the association of systemic chemotherapy with wound care by hydrogel helped in resolving the superficial skin infections, preventing a potential source of sepsis. We therefore conclude that this type of topical wound treatment may be of help in the management of ulcerative lesions in patients with lymphoproliferative disease associated with immunosuppression.
References
1. Bodey GP, Bolivar R, Fainstain V. Infectious complications in leukemic patients. Semin Hematol 1982; 19: 193-2262. Pizzo PA. Infectious complications in the child with cancer. I,II, III. J Pediatr 1981; 98: 341-354, 513-523, 524-530
3. Wolfson JS, Sober AJ, Rubin RH. Dermatologic manifestations of infections in immunocompromised patients. Medicine 1985; 64: 115-133
4. Dalton JA,Yag-Howard C, Messina JL, et al. Cutaneous T-cell lymphoma. Int J Dermatol 1997; 36: 801-809
5. Kim YH, Hoppe RT. Mycosis fungoides and the Sezary syndrome. Semin Oncol 1999; 26: 276-289
6. Axelrod PI, Lorber B,Vonderheid EC. Infections complicating mycosis fungoides and Sezary syndrome. JAMA 1992; 267: 1354-1358
7. Posner LE, Fossieck BE Jr, Eddy JL, Bunn PA Jr. Septicemic complications of the cutaneous T-cell lymphomas. Am J Med 1981; 71: 210-216
8. Heald P, Yan SL, Edelson R. Profound deficiency in normal circulating T cells in erythrodermic cutaneous T-cell lymphoma. Arch Dermatol 1994; 130: 198-203.
9. Yawalkar N, Ferenczi K, Jones DA, Yamanaka K, Suh KY, Sadat S, Kupper TS. Profound loss of T-cell receptor repertoire complexity in cutaneous T-cell lymphoma. Blood 2003 1; 102: 4059-4066
10. Carbia SG, Hochman A, Dei-Cas I, Lagodin C, Melgar A, Allevato M, Woscoff A. Ulcerated cutaneous lymphoma. 10 years of experience. Medicina (B Aires). 2000;60(5 Pt 1):565-569
© 2006 Dermatology Online Journal

