Malignant transformation of donovanosis (granuloma inguinale) in a HIV-postive patient
Published Web Location
https://doi.org/10.5070/D36gd7z92mMain Content
Malignant transformation of donovanosis (granuloma inguinale) in a HIV-postive patient
Kabir Sardana1, Vijay K Garg2 Pooja Arora3, Neeta Khurana4
Dermatology Online Journal 14 (9): 8
Maulana Azad Medical College, Delhi, India1. Asst. Prof. Dermatology. kabirijdvl@gmail.com
2. Prof. & HOD, Dermatology
3. Junior Resident Dermatology
4. Assoc. Prof. Pathology
Abstract
Donovanosis is endemic in various parts of the world. Although donovanosis has been reported to co-exist with the human immunodeficiencyvirus (HIV) infection, to our knowledge there have been no reports of co-existent squamous cell carcinoma (SCC) in an HIV positive patient. In our case the patient did not respond to treatment for donovanosis and on biopsy we realized that the patient had coexistent SCC, which is hitherto unreported with granuloma inguinale.
Introduction
Donovanosis is caused by Calymmatobacterium granulomatis. It is endemic in new Guinea, the Caribbean, South Africa, Southeast Asia, Australia, Brazil and southern India [1, 2, 3, 4]. In Pondicherry, South India, between 1993 and 1997, donovanosis constituted 14 percent of genital ulcer disease patients, 15 percent of whom were HIV positive. However, in North India, the incidence declined significantly in the 1990s [2, 5]. Although donovanosis has been reported to coexist with HIV infection, to our knowledge there have been no reports of co-existent SCC in an HIV positive patient.
Case report
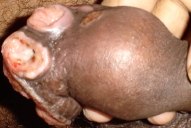
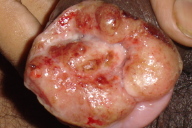
A 21-year-old unmarried male, presented with multiple asymptomatic penile ulcerations of one-year duration. He denied drug abuse and was heterosexually promiscuous. The lesions started initially as painless small raised nodules over the prepuce (Fig. 1a) and gradually increased in size over a period of four months and subsequently broke down to leave behind red, raw, painless ulcers (Fig. 1b). The patient continued to develop similar lesions that followed a similar course. Examination revealed multiple non-tender ulcers with raised, rolled out margins and exuberant, pale red, granulation tissue that showed bleeding on touch and were present over the prepuce obscuring the glans and urinary meatus (Figs. 1a & 1b). Three non-tender firm papulo-nodules were seen adjacent to the ulcer.
 |  |
| Figure 1c | Figure 1d |
|---|---|
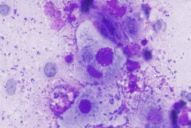
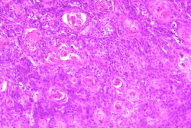
| Figure 1c. Giemsa smear showing intracellular basophilic Donovan bodies (x40) Figure 1d. Histology revealing showed nests of squamous cells and keratin pearls (x40 H&E) | |
A tissue smear from the ulcer margin stained by rapid Giemsa showed Donovan bodies within mononuclear cells (Fig. 1c). Lesional fine needle aspiration cytology (FNAC) showed multiple atypical squamous cells, occasional granulomas, and inflammatory cells in the background. Biopsy from the ulcer edge showed nests of squamous cells with a high degree of mitosis (Fig. 1d). Fine needle aspiration cytology from inguinal lymph nodes showed reactive lymphoid hyperplasia. Serologic testing for HIV type I was positive, CD4+ lymphocyte count was 345 cells/mm³. A repeat biopsy was done and it confirmed the diagnosis of squamous cell carcinoma (moderately differentiated). The patient was treated with highly active antiretroviral therapy (HAART), doxycycline (100mg BID for 3 weeks), and cotrimoxazole double-strength (BID for 21 days).
In view of the histology report total penectomy with bilateral modified inguinal lymph node dissection was performed. The repeat histology from the dissected specimen confirmed the presence of squamous cell carcinoma. The patient is now being planned for post operative chemo-radiotherapy.
Discussion
Donovanosis is a chronic cause of genital ulceration, the incidence of which is decreasing significantly in developed and developing countries [3, 4, 5]. The pathologic agent is Calymmatobacterium granulomatis, a gram negative, encapsulated, facultative, obligate intracellular, pleomorphic bacillus. More than 70 percent of cases occur between the ages of 20 and 40 years [4, 5]; it most commonly afflicts patients in the 3rd decade of life [6]. The infectivity rate among sexual partners ranges from 0.4 percent to 52 percent [5, 7, 8, 9, 10]. After an incubation period of eight days to twelve weeks (most commonly two weeks), single or multiple subcutaneous papules or nodules grow and erode to form painless, beefy ulcerations with friable, clean bases and distinct, rolled, raised margins [3, 4]. Without treatment, lesions may become confluent and may cause progressive mutilation and destruction of local tissue [3, 4, 10]. This was typically seen in our case.
Genital ulceration leads to 4.7 times higher risk of HIV transmission after sexual exposure to an HIV infected person [1]. Coinfection with HIV is likely to persist for a longer duration and thus require more intensive and prolonged antibiotic therapy compared to HIV negative patients with donovanosis (Table 1) [11, 12]. Our patient also received two antibiotic therapies in view of the concomitant HIV positivity. A prospective case-control study of 50 Indian patients showed that the ulcer size and clinical presentation were not significantly different in HIV positive and HIV negative patients [10]. There was greater tissue destruction and significantly longer time to healing 29 (25.7 days) versus 16.8 days [10] (Table 1). A summary of features of concomitant HIV with donovaniosis is enlisted (Table 1) and highlights the rarity of SCC in this setting [12].
Carcinoma is the most serious complication of donovanosis but is relatively rare, 0.25 percent in Rajam and Ranjiah's series [8]. Palpable inguinal lymphadenopathy is present in 58 percent of patients with carcinoma of the penis. Of these, 45 percent have cancer in the nodes and the remainder have inflammatory lymphadenopathy secondary to infection of the primary tumor. A recent study supporting our observation revealed that in cases of penile cancer 10/25 FNAC from the inguinal region were negative [2, 13]. This could explain the lack of histological involvement of the lymph node in our case. Unlike patients with superficially invasive (<2 cm) or well differentiated tumors, those with high grade squamous cell carcinoma, extensive (>75%) penile shaft involvement, and a flat growth pattern have an increased incidence of involvement of inguinal lymph nodes [13].
Fine needle aspiration cytology and two repeat biopsies from the ulcer edge confirmed the diagnosis of SCC. The abundant Donovan bodies and typical clinical morphology led to the diagnosis of donovanosis with SCC. This is the first case report of an HIV positive patient with donovanosis undergoing malignant transformation.
References
1. Manders MD, Baxter JD. Granuloma-inguinale and HIV: A unique presentation and novel treatment regimen. J Am Acad Dermatol 1997; 37:494-6. PubMed2. DM Thappa, Senthilkumar M, Laxmisha C. Malignant transformation of Donovanosis. Indian J Sex Transm. Dis 2004;l25 (2),77.
3. Goens JL, Schwartz RP, de Woy K. Mucocutaneous manifestations of chancroid, lymphogranuloma venereum and granuloma inguinale. Am Fam Physician 1994;49:415-25. PubMed
4. O'Farlell N. Donovanosis. Sex Transm Infect 2002; 78:452-7. PubMed
5. Lal S, Nicholas C. Epidemiological and clinical features in 165 cases of Granuloma inguinale. Br. J.Vener Dis.1970;46:461-3. PubMed
6. Mitchell KM, Roberts AN, Williams VM, et al. Donovanosis in western Austialia Genitourin Med 1986;62:191-5. PubMed
7. Seema JS. Granuloma Venerium: a problem in Guntur. Indian J Dermatol Venereol Leprol 1969;5:45-52.
8. Rajam and Rangaiah. Donovanosis, WHO Monograph Series 1954;Number1( 24): 1-72
9. Anandam K. Study of granuloma venereum. Indian J Dermatol Venereol Leprol 1979; 45: 323-332
10. Jamkhedkas PP, Hira SK, Shroff HJ, et al. Clinico epidemiologic features of granuloma inguinal in the era of acquired immunodeficiency syndrome. Sex Transm Dis 1998;25:196-200. PubMed
11. CJ Sanders. Extragenital donovanosis in a patient with AIDS. Sex. Transm. Inf. 1998;74;142-143 PubMed
12. Sardana K , Sehgal VN. Genital ulcer disease and human immuno deficiency virus: a focus. Int J Dermatol 2005;44:391-405. PubMed
13. Saisorn I, Lawrentschuk N, Leewansangtong S, Bolton DM. Fine-needle aspiration cytology predicts inguinal lymph node metastasis without antibiotic pretreatment in penile carcinoma .BJU Int. 2006 ;97(6):1225-8. PubMed
© 2008 Dermatology Online Journal