Colocalized nevus depigmentosus and lentigines with underlying breast hypoplasia: A case of reverse mutation?
Published Web Location
https://doi.org/10.5070/D33h4111gcMain Content
Colocalized nevus depigmentosus and lentigines with underlying breast hypoplasia: A case of reverse mutation?
Rachna Jagia1, Vibhu Mendiratt 1, RV Koranne 1, Kabir Sardana1, Premanshu Bhushan 1, and RS Solanki2
Dermatology Online Journal 10 (1): 12
From the Departments of Dermatology & Sexually Transmitted Diseases1, and Radiodiagnosis2, Lady Hardinge Medical College & Associated S.S.K. & K.S.C. Hospitals, New Delhi, INDIA. kabir_sardana1@rediffmail.com
Abstract
Nevus depigmentosus (ND) is classically defined as a congenital nonprogressive hypopigmented macule, stable in size and distribution. There have been many reports of colocalization of ND and lentigines. We describe development of multiple lentigines over ND in a 9-year-old girl along with hypoplasia of the underlying breast. The case is being reported to highlight the phenotypic manifestation of reverse mutation and the coincidental breast hypoplasia that has not been reported before.
Clinical summary
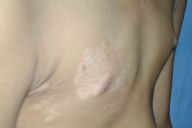
A 9-year-old girl had multiple hypopigmented patches first noticed at age 2 years, with overlying hyperpigmented spots since age 3. Examination revealed 7 oval hypopigmented macules 1 × 1cm to 3 × 5 cm in size with serrated, irregular borders involving the right mammary, upper scapular, and axillary regions. The lesions were without poliosis or whirling (fig. 1). The lesions followed the Blaschko lines with a sharp midline demarcation. There was an offwhite accentuation on Wood's lamp examination. Multiple colocalized hyperpigmented macules (1-5 mm diameter) suggestive of lentigines were present. At a 2-year followup, the number and color of lentigines remained unchanged but there was hypoplasia of the right breast although the nipple and areola were normal. Psychomotor and musculoskeletal development were normal. There was no history of breast lump or trauma to the area and the family history was non-contributory.
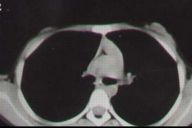
Histological examination of a hypopigmented macule revealed a normal number of melanocytes on S-100 staining, consistent with nevus depigmentosus. The colocalized hyperpigmented macules demonstrated an increase in the melanin content within the keratinocytes of the basal layer, with focal areas of increase in the number of melanocytes. X-ray of the lumbosacral spine revealed spina bifida of S-1 vertebra with normal intervertebral disc spaces. There was no abnormality in electroencephalography or in computerized tomogarphic (CT) scan of head. A CT scan of the chest showed hypoplasia of underlying breast tissue compared to left side, with normal underlying muscle and bones (fig 2).
Discussion
The commonly used clinical diagnostic criteria for nevus depigmentosus (ND) are as follows [1, 2, 3]:
- Leukoderma present at birth or of an early onset
- No alteration in distribution of leukoderma throughout life
- No alteration in texture or change of sensation in the affected area
- Absence of hyperpigmented border
Lee et al. found that majority (92.5 %) of ND present before age 3 years and have serrated irregular borders (77.4 %) [4]. ND is generally classified into isolated, segmental, and whorled types [4]. Wood's lamp examination shows an off-white accentuation in ND (compared to chalky-white in vitiligo). Histopathologically, the numbers of melanocytes are normal or decreased [1, 5, 6] but DOPA reactivity is consistently reduced [1]. Melanosomes are usually normal in size, shape, and internal structure[1], but can be decreased in number, heteromorphic, aggregated in melanocytes, or located in membrane bound aggregates [4].
ND is not usually associated with other abnormalities but there are case reports of associated seizures, mental retardation, unilateral limb hypertrophy, atopic dermatitis, and abnormal systemic features [7]. All of the above are cases of neurocutaneous disorders labeled as ND but were perhaps hypomelanosis of Ito (HOI). The diagnostic criteria differentiating whorled ND from HOI are vague [8]. However, cases of HOI without extracutaneous anomalies are often categorized in the literature as ND, whereas cases of ND associated with extracutaneous anomalies are usually categorized as HOI [7]. Probably these two entities represent a group of heterogeneous diseases with genetic mosaicism as the common, underlying factor [9]. In the future, patients currently diagnosed with either of these disorders might simply be categorized as having nevoid hypopigmentation secondary to somatic mosaicism [9]. Figure 3 presents the review of the reported cases of ND with lentigines.
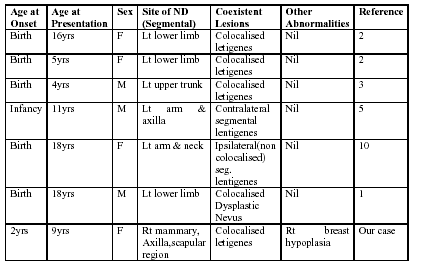
|
| Figure 3 |
|---|
| A summary of case of nevus depigmentosus and lentigines |
Colocalized defects may have different origins. Field defect theory describes the phenomenon of polytopic malformations. These result from genetic defects manifested during blastogenesis because of the spacial contiguity, hierarchy, and close timing of morphogenic events [11]. Examples of abnormalities explained by this effect include four case reports of colocalized lentigines [1, 2]. Alternatively, two reports of simultaneous but separate segmental lentigines have been explained by the twin-spotting theory [5]. This theory explains the existence of two distinct patches of mutant skin that are genetically different from each other and from the surrounding normal skin. In this case, the involved individual is heterozygous for two different mutations on two homologous chromosomes. Somatic recombination can result in two daughter cells, homozygous for either mutation. These two daughter cells may then produce two different mutant clones that form paired mutant patches [12]. Colocalization of lentigines can be regarded as a different form of repigmentation resulting from reverse mutation in one of the genes involved in pigmentation [1]. This would restore pigment function, although perhaps in a less tightly controlled fashion [2], the underlying breast atrophy could also be a manifestation of a field defect, since the skin and the breast tissue have a common embryological origin.
References
1. Shim JH, Seo SJ, Song KY, Hong CK. Development of multiple pigmented nevi within segmental nevus depigmentosus. J Korean Med Sci. 2002 Feb;17(1):133-6. PubMed2. Bolognia JL, Lazova R, Watsky K. The development of lentigines within segmental achromic nevi. J Am Acad Dermatol. 1998 Aug;39(2 Pt 2):330-3. PubMed
3. Khumalo NP, Huson S, Burge S. Development of lentigines within naevoid hypopigmentation. Br J Dermatol. 2001 Jan;144(1):188-9. No abstract available. PubMed
4. Lee HS, Chun YS, Hann SK. Nevus depigmentosus: clinical features and histopathologic characteristics in 67 patients. J Am Acad Dermatol. 1999 Jan;40(1):21-6. PubMed
5. Alkemade H, Juhlin L. Unilateral lentiginosis with nevus depigmentosus on the other side. J Am Acad Dermatol. 2000 Aug;43(2 Pt 2):361-3. PubMed
6. Happle R, Barbi G, Eckert D, Kennerknecht I. "Cutis tricolor": congenital hyper- and hypopigmented macules associated with a sporadic multisystem birth defect: an unusual example of twin spotting? J Med Genet. 1997 Aug;34(8):676-8. PubMed
7. Nehal KS, PeBenito R, Orlow SJ. Analysis of 54 cases of hypopigmentation and hyperpigmentation along the lines of Blaschko. Arch Dermatol. 1996 Oct;132(10):1167-70. PubMed
8. Ruiz-Maldonado R, Toussaint S, Tamayo L, Laterza A, del Castillo V. Hypomelanosis of Ito: diagnostic criteria and report of 41 cases. Pediatr Dermatol. 1992 Mar;9(1):1-10. PubMed
9. Loomis CA. Linear hypopigmentation and hyperpigmentation, including mosaicism. Semin Cutan Med Surg. 1997 Mar;16(1):44-53. Review. PubMed
10. Baba M, Akcali C, Seckin D, Happle R. Segmental lentiginosis with ipsilateral nevus depigmentosus: another example of twin spotting? Eur J Dermatol. 2002 Jul-Aug;12(4):319-21. PubMed
11. Martinez-Frias ML, Frias JL, Opitz JM. Errors of morphogenesis and developmental field theory. Am J Med Genet. 1998 Apr 1;76(4):291-6. PubMed
12. Happle R. Dohi Memorial Lecture. New aspects of cutaneous mosaicism. J Dermatol. 2002 Nov;29(11):681-92. PubMed
© 2004 Dermatology Online Journal